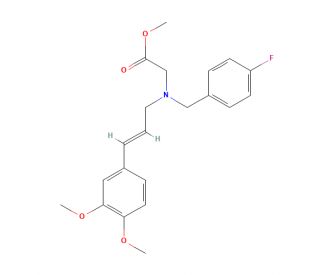
Molecular structure of Abamine, CAS Number: 729612-64-0
Abamine (CAS 729612-64-0)
Alternate Names:
N-[(2E)-3-(3,4-Dimethoxyphenyl)-2-propen-1-yl]-N-[(4-fluorophenyl)methyl]-glycine Methyl Ester
CAS Number:
729612-64-0
Molecular Weight:
373.42
Molecular Formula:
C21H24FNO4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Abamine is a 9-Cis-Epoxycarotenoid dioxygenase (NCED) inhibitor which is a key regulatory enzyme of the abscisic acid biosynthesis pathway. Abamine is a synthetic molecule designed to replicate the properties and functions of natural compounds. Abamine finds use in scientific research for investigating various biochemical and physiological processes. Abamine has proven instrumental in exploring enzyme kinetics, unraveling protein structures, and deciphering cell signaling pathways.
Abamine (CAS 729612-64-0) References
- A novel inhibitor of 9-cis-epoxycarotenoid dioxygenase in abscisic acid biosynthesis in higher plants. | Han, SY., et al. 2004. Plant Physiol. 135: 1574-82. PMID: 15247398
- Control of nodule number by the phytohormone abscisic Acid in the roots of two leguminous species. | Suzuki, A., et al. 2004. Plant Cell Physiol. 45: 914-22. PMID: 15295075
- Suppression of root nodule formation by artificial expression of the TrEnodDR1 (coat protein of White clover cryptic virus 1) gene in Lotus japonicus. | Nakatsukasa-Akune, M., et al. 2005. Mol Plant Microbe Interact. 18: 1069-80. PMID: 16255246
- A 9-cis-epoxycarotenoid dioxygenase inhibitor for use in the elucidation of abscisic acid action mechanisms. | Kitahata, N., et al. 2006. Bioorg Med Chem. 14: 5555-61. PMID: 16682205
- Sesquiterpene-like inhibitors of a 9-cis-epoxycarotenoid dioxygenase regulating abscisic acid biosynthesis in higher plants. | Boyd, J., et al. 2009. Bioorg Med Chem. 17: 2902-12. PMID: 19269833
- Enhanced nodulation and nitrogen fixation in the abscisic acid low-sensitive mutant enhanced nitrogen fixation1 of Lotus japonicus. | Tominaga, A., et al. 2009. Plant Physiol. 151: 1965-76. PMID: 19776164
- Effect of abscisic acid on symbiotic nitrogen fixation activity in the root nodules of Lotus japonicus. | Tominaga, A., et al. 2010. Plant Signal Behav. 5: 440-3. PMID: 20118670
- Study of 'Redhaven' peach and its white-fleshed mutant suggests a key role of CCD4 carotenoid dioxygenase in carotenoid and norisoprenoid volatile metabolism. | Brandi, F., et al. 2011. BMC Plant Biol. 11: 24. PMID: 21269483
- Substrate-directable Heck reactions with arenediazonium salts. The regio- and stereoselective arylation of allylamine derivatives and applications in the synthesis of naftifine and abamines. | Prediger, P., et al. 2011. J Org Chem. 76: 7737-49. PMID: 21877731
- Plastid stromules are induced by stress treatments acting through abscisic acid. | Gray, JC., et al. 2012. Plant J. 69: 387-98. PMID: 21951173
- Plastid Located WHIRLY1 Enhances the Responsiveness of Arabidopsis Seedlings Toward Abscisic Acid. | Isemer, R., et al. 2012. Front Plant Sci. 3: 283. PMID: 23269926
- Target sites for chemical regulation of strigolactone signaling. | Nakamura, H. and Asami, T. 2014. Front Plant Sci. 5: 623. PMID: 25414720
- Abscisic acid-mediated modifications of radial apoplastic transport pathway play a key role in cadmium uptake in hyperaccumulator Sedum alfredii. | Tao, Q., et al. 2019. Plant Cell Environ. 42: 1425-1440. PMID: 30577078
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Abamine, 25 mg | sc-479391 | 25 mg | $425.00 | |||
Abamine, 50 mg | sc-479391A | 50 mg | $800.00 | |||
Abamine, 100 mg | sc-479391B | 100 mg | $1348.00 |
