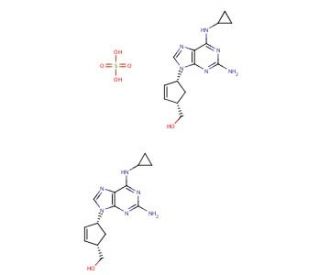
Abacavir Sulfate (CAS 188062-50-2)
QUICK LINKS
Abacavir sulfate is the sulfate salt form of abacavir, a synthetic nucleoside analogue within the guanine derivatives class. This compound is extensively used in scientific research to study the mechanisms of nucleoside reverse transcriptase inhibitors (NRTIs) and their effects on viral replication, particularly focusing on HIV-1. The primary mechanism of action of abacavir sulfate involves its intracellular conversion to the active triphosphate form, carbovir triphosphate. This active form mimics the natural nucleoside substrates utilized by the viral reverse transcriptase enzyme. Carbovir triphosphate competes with the natural deoxyguanosine triphosphate (dGTP) for incorporation into the viral DNA chain by reverse transcriptase. Once incorporated, it terminates the DNA chain because it lacks a 3′-OH group, necessary to form the next phosphodiester bond in the DNA chain. This termination effectively inhibits the replication of the HIV virus by halting DNA synthesis. In research settings, abacavir sulfate is utilized to study the processes involving reverse transcription and to understand how nucleoside analogues can inhibit this critical step in the HIV life cycle. Researchers use this compound to examine the kinetics of viral reverse transcriptase inhibition and explore how mutations in the reverse transcriptase enzyme affect the interaction with nucleoside analogues. Additionally, abacavir sulfate is employed in studies focused on the pharmacokinetics and dynamics of nucleoside analogues. These studies investigate how structural modifications influence the efficacy and metabolism of these compounds, enhancing understanding of their behavior and developing new strategies to optimize their performance.
Abacavir Sulfate (CAS 188062-50-2) References
- Abacavir-lamivudine-zidovudine vs indinavir-lamivudine-zidovudine in antiretroviral-naive HIV-infected adults: A randomized equivalence trial. | Staszewski, S., et al. 2001. JAMA. 285: 1155-63. PMID: 11231744
- Development and validation of a reverse-phase liquid chromatographic method for assay and related substances of abacavir sulfate. | Seshachalam, U., et al. 2007. J Sep Sci. 30: 28-34. PMID: 17313138
- Separation and characterization of forced degradation products of abacavir sulphate by LC-MS/MS. | Rao, RN., et al. 2011. J Pharm Biomed Anal. 54: 279-85. PMID: 20869185
- Stress Degradation Behavior of Abacavir Sulfate and Development of a Suitable Stability-Indicating UHPLC Method for the Determination of Abacavir, its Related Substances, and Degradation Products. | Vukkum, P., et al. 2012. Sci Pharm. 80: 903-21. PMID: 23264939
- Stability behaviour of antiretroviral drugs and their combinations. 5: Characterization of novel degradation products of abacavir sulfate by mass and nuclear magnetic resonance spectrometry. | Kurmi, M., et al. 2017. J Pharm Biomed Anal. 134: 372-384. PMID: 27889293
- 1592U89, a novel carbocyclic nucleoside analog with potent, selective anti-human immunodeficiency virus activity. | Daluge, SM., et al. 1997. Antimicrob Agents Chemother. 41: 1082-93. PMID: 9145874
- Abacavir. | Foster, RH. and Faulds, D. 1998. Drugs. 55: 729-36; discussion 737-8. PMID: 9585869
- Microspheres of carboxymethyl guar gum for in vitro release of abacavir sulfate: preparation and characterization | Sullad, A. G., Manjeshwar, L. S., & Aminabhavi, T. M. 2011. Journal of Applied Polymer Science. 122(1): 452-460.
- Novel semi-interpenetrating microspheres of dextran-grafted-acrylamide and poly (vinyl alcohol) for controlled release of abacavir sulfate | Sullad, A. G., Manjeshwar, L. S., & Aminabhavi, T. M. 2011. Industrial & engineering chemistry research. 50(21): 11778-11784.
- Factorial design for development of a high-performance thin-layer chromatography method for the simultaneous estimation of abacavir sulfate, lamivudine hydrochloride, and dolutegravir sodium | Dave, K., & Desai, S. 2018. JPC-Journal of Planar Chromatography-Modern TLC. 31(6): 489-495.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Abacavir Sulfate, 5 mg | sc-207238 | 5 mg | $146.00 | |||
Abacavir Sulfate, 25 mg | sc-207238A | 25 mg | $244.00 |
