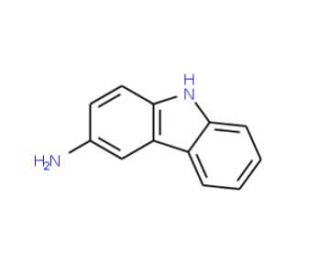
Molecular structure of 9H-Carbazol-3-ylamine, CAS Number: 6377-12-4
9H-Carbazol-3-ylamine (CAS 6377-12-4)
Alternate Names:
3-Aminocarbazole
CAS Number:
6377-12-4
Molecular Weight:
182.23
Molecular Formula:
C12H10N2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
9H-Carbazol-3-ylamine, also referred to as 9H-carbazole, acts as a starting material in the creation of other heterocyclic compounds like quinolones and carbazoles. The compound is also used in the synthesis of various bioactive compounds. Furthermore, 9H-Carbazol-3-ylamine is used as a ligand in coordination chemistry. While the action mechanism of 9H-Carbazol-3-ylamine is not completely understood, it is thought to play a role in inhibiting certain enzymes, like cytochrome P450, which are part of the metabolism of various xenobiotic compounds. Additionally, there is evidence of 9H-Carbazol-3-ylamine interacting with DNA, which suggests a possible role in gene expression regulation.
9H-Carbazol-3-ylamine (CAS 6377-12-4) References
- Synthesis and evaluation of cytotoxic activities of new guanidines derived from carbazoles. | Caruso, A., et al. 2014. Bioorg Med Chem Lett. 24: 467-72. PMID: 24374274
- Mutagenicity of aminocarbazoles and nitrocarbazoles. | LaVoie, EJ., et al. 1981. Mutat Res. 90: 337-44. PMID: 7038457
- Mutagenicity of nitro- and amino-substituted carbazoles in Salmonella typhimurium. I. Monosubstituted derivatives of 9H-carbazole. | André, V., et al. 1993. Mutat Res. 299: 63-73. PMID: 7679194
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
9H-Carbazol-3-ylamine, 500 mg | sc-326119 | 500 mg | $363.00 |
