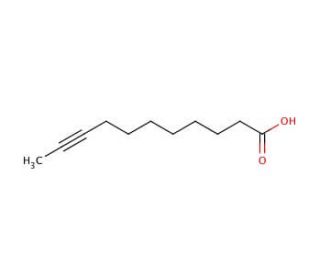
9-Undecynoic acid (CAS 22202-65-9)
QUICK LINKS
9-Undecynoic acid is a fatty acid derivative that functions as an inhibitor of various enzymes involved in lipid metabolism. It exerts its mechanism of action by interfering with the activity of enzymes such as stearoyl-CoA desaturase and fatty acid synthase, which are used for the biosynthesis of fatty acids. By inhibiting these enzymes, 9-Undecynoic acid disrupts the normal process of lipid synthesis, ultimately leading to a reduction in the production of important cellular components such as triglycerides and phospholipids. This disruption in lipid metabolism can have downstream effects on cellular processes and signaling pathways, making 9-Undecynoic acid useful for studying the regulation of lipid metabolism and its impact on cellular function. Its mechanism of action at the molecular level provides insights into the network of metabolic pathways.
9-Undecynoic acid (CAS 22202-65-9) References
- Ring-closing alkyne metathesis. Application to the total synthesis of sophorolipid lactone. | Fürstner, A., et al. 2000. J Org Chem. 65: 8758-62. PMID: 11112600
- Analysis of cigarette smoke condensates by comprehensive two-dimensional gas chromatography/time-of-flight mass spectrometry I acidic fraction. | Lu, X., et al. 2003. Anal Chem. 75: 4441-51. PMID: 14632048
- Polyurethane networks from fatty-acid-based aromatic triols: synthesis and characterization. | Lligadas, G., et al. 2007. Biomacromolecules. 8: 1858-64. PMID: 17472338
- Potent in vitro antifungal activities of naturally occurring acetylenic acids. | Li, XC., et al. 2008. Antimicrob Agents Chemother. 52: 2442-8. PMID: 18458131
- Catalysis-based and protecting-group-free total syntheses of the marine oxylipins hybridalactone and the ecklonialactones A, B, and C. | Hickmann, V., et al. 2011. J Am Chem Soc. 133: 13471-80. PMID: 21780792
- Selective reduction of carboxylic acids to aldehydes catalyzed by B(C6F5)3. | Bézier, D., et al. 2013. Org Lett. 15: 496-9. PMID: 23317512
- Lactones in the Synthesis of Prostaglandins and Prostaglandin Analogs. | Tănase, C., et al. 2021. Int J Mol Sci. 22: PMID: 33557221
- Design of Chemically Stable Organic Perovskite Quantum Dots for Micropatterned Light-Emitting Diodes through Kinetic Control of a Cross-Linkable Ligand System. | Lee, H., et al. 2021. Adv Mater. 33: e2007855. PMID: 33938035
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
9-Undecynoic acid, 1 g | sc-227147 | 1 g | $47.00 |
