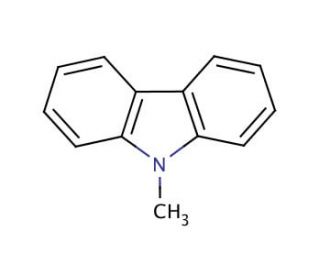
9-Methylcarbazole (CAS 1484-12-4)
QUICK LINKS
9-Methylcarbazole is a chemical compound that functions as a fluorescent dye in research and development applications. It is utilized to label and visualize specific molecules or structures within cells and tissues. The compound′s mechanism of action involves binding to target molecules or structures, emitting fluorescence when excited by specific wavelengths of light. This allows researchers to track and study the localization and movement of the labeled molecules or structures in various experimental assays. 9-Methylcarbazole can be used to detect and quantify specific biomolecules in biochemical assays, contributing to the understanding of molecular interactions and cellular processes. Its ability to emit fluorescence upon binding to target molecules may be useful for visualizing and analyzing biological samples in research and development applications.
9-Methylcarbazole (CAS 1484-12-4) References
- Design, synthesis, and structure-activity correlations of novel dibenzo[b,d]furan, dibenzo[b,d]thiophene, and N-methylcarbazole clubbed 1,2,3-triazoles as potent inhibitors of Mycobacterium tuberculosis. | Patpi, SR., et al. 2012. J Med Chem. 55: 3911-22. PMID: 22449006
- Spectral and thermodynamic properties for the exciplexes of N-alkyl carbazoles with dicyanobenzenes in THF. | Asim, S., et al. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 118: 138-45. PMID: 24051282
- Novel 3-substituted N-methylcarbazole-imidazolium salt derivatives: Synthesis and cytotoxic activity. | Li, YH., et al. 2018. Chem Biol Drug Des. 92: 1206-1213. PMID: 29430875
- Magnetic solid phase extraction of heterocyclic aromatic hydrocarbons from environmental water samples with multiwalled carbon nanotube modified magnetic polyamido-amine dendrimers prior to gas chromatography-triple quadrupole mass spectrometer. | Zhou, Q., et al. 2021. J Chromatogr A. 1639: 461921. PMID: 33524931
- Graphene oxide modified magnetic polyamidoamide dendrimers based magnetic solid phase extraction for sensitive measurement of polycyclic aromatic hydrocarbons. | Tong, Y., et al. 2022. Chemosphere. 296: 134009. PMID: 35189186
- 18O studies of the peroxidase-catalyzed oxidation of N-methylcarbazole. Mechanisms of carbinolamine and carboxaldehyde formation. | Kedderis, GL., et al. 1986. J Biol Chem. 261: 15910-4. PMID: 3782097
- Mutagenicity of substituted carbazoles in Salmonella typhimurium. | LaVoie, EJ., et al. 1982. Mutat Res. 101: 141-50. PMID: 7048081
- Metabolism of N-methylcarbazole by rat lung microsomes. | Ibe, BO. and Raj, JU. 1994. Exp Lung Res. 20: 207-22. PMID: 7925139
- Chemiexcitation in the peroxidative metabolism of N-methylcarbazole: mechanistic implications. | de Melo, MP. and Cilento, G. 1994. Photochem Photobiol. 59: 677-82. PMID: 8066126
- Inactivation of purified rat liver cytochrome P-450 2B1 and rabbit liver cytochrome P-450 2B4 by N-methylcarbazole. | Kuemmerle, SC., et al. 1994. Drug Metab Dispos. 22: 343-51. PMID: 8070309
- The mechanism of stimulation of NADPH oxidation during the mechanism-based inactivation of cytochrome P450 2B1 by N-methylcarbazole: redox cycling and DNA scission. | Shen, T. and Hollenberg, PF. 1994. Chem Res Toxicol. 7: 231-8. PMID: 8199313
- Bioassay of quinoline, 5-fluoroquinoline, carbazole, 9-methylcarbazole and 9-ethylcarbazole in newborn mice. | Weyand, EH., et al. 1993. Food Chem Toxicol. 31: 707-15. PMID: 8225128
- Microbial models of mammalian metabolism: involvement of cytochrome P450 in the N-demethylation of N-methylcarbazole by Cunninghamella echinulata. | Yang, W., et al. 1993. Xenobiotica. 23: 973-82. PMID: 8291265
- The formation of an N-hydroxymethyl intermediate in the N-demethylation of N-methylcarbazole in vivo and in vitro. | Gorrod, JW. and Temple, DJ. 1976. Xenobiotica. 6: 265-74. PMID: 936646
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
9-Methylcarbazole, 5 g | sc-227145 | 5 g | $136.00 |
