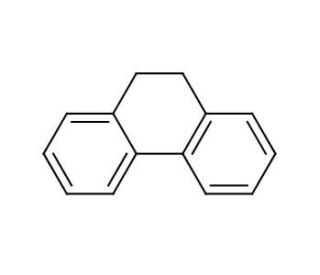
9,10-Dihydrophenanthrene (CAS 776-35-2)
QUICK LINKS
9,10-Dihydrophenanthrene is a polycyclic hydrocarbon of interest primarily in organic and environmental chemistry research. Its structure, representing a partially saturated version of phenanthrene, makes it a useful model compound for studying the hydrogenation and aromatic stabilization energies of polycyclic aromatic hydrocarbons (PAHs). In environmental studies, it is often used as a non-aromatic analogue to understand the persistence and degradation pathways of PAHs in the soil and water, which are critical for assessing environmental contamination and remediation strategies. Additionally, chemists utilize 9,10-Dihydrophenanthrene to investigate the mechanisms of electrophilic aromatic substitution reactions, providing valuable insights into the synthesis of complex organic molecules.
9,10-Dihydrophenanthrene (CAS 776-35-2) References
- Photophysical and photochemical processes of 9,10-dihydro-9-silaphenanthrene derivatives: photochemical formation and electronic structure of 9-silaphenanthrenes. | Hiratsuka, H., et al. 2006. J Phys Chem A. 110: 3868-74. PMID: 16539407
- 9, 10-Dihydrophenanthrene derivatives from Pholidota yunnanensis and scavenging activity on DPPH free radical. | Guo, XY., et al. 2007. J Asian Nat Prod Res. 9: 165-74. PMID: 17454314
- Nickel-catalyzed [2+2+2] cycloaddition of arynes and an unactivated alkene: synthesis of 9,10-dihydrophenanthrene derivatives. | Saito, N., et al. 2009. Chem Commun (Camb). 4284-6. PMID: 19585048
- A new 9,10-dihydrophenanthrene and cell proliferative 3,4-δ-dehydrotocopherols from Stemona tuberosa. | Kil, YS., et al. 2015. Molecules. 20: 5965-74. PMID: 25854758
- A new 9,10-dihydrophenanthrene from Dendrobium moniliforme. | Zhao, N., et al. 2016. Nat Prod Res. 30: 174-9. PMID: 26132274
- New phenanthrene and 9, 10-dihydrophenanthrene derivatives from the stems of Dendrobium officinale with their cytotoxic activities. | Zhao, GY., et al. 2018. J Nat Med. 72: 246-251. PMID: 29063360
- A new phenanthrene and a new 9,10-dihydrophenanthren from Bulbophyllum retusiusculum. | Sun, J., et al. 2018. Nat Prod Res. 32: 2447-2451. PMID: 29284299
- Isolation, Structural Elucidation, Optical Resolution, and Antineuroinflammatory Activity of Phenanthrene and 9,10-Dihydrophenanthrene Derivatives from Bletilla striata. | Zhou, D., et al. 2019. J Nat Prod. 82: 2238-2245. PMID: 31415170
- Phenanthrene, 9,10-dihydrophenanthrene and bibenzyl enantiomers from Bletilla striata with their antineuroinflammatory and cytotoxic activities. | Sun, MH., et al. 2021. Phytochemistry. 182: 112609. PMID: 33326906
- Discovery of 9,10-dihydrophenanthrene derivatives as SARS-CoV-2 3CLpro inhibitors for treating COVID-19. | Zhang, JW., et al. 2022. Eur J Med Chem. 228: 114030. PMID: 34883292
- Rational identification of small molecules derived from 9,10-dihydrophenanthrene as potential inhibitors of 3CLpro enzyme for COVID-19 therapy: a computer-aided drug design approach. | Daoui, O., et al. 2022. Struct Chem. 33: 1667-1690. PMID: 35818588
- The anti-SARS-CoV-2 activity of novel 9, 10-dihydrophenanthrene derivatives: an insight into molecular docking, ADMET analysis, and molecular dynamics simulation. | Yamari, I., et al. 2023. Sci Afr. 21: e01754. PMID: 37332393
- Synthesis and antimalarial evaluation of 9,10-dihydrophenanthrene amino alcohols. | Dey, AS. and Neumeyer, JL. 1974. J Med Chem. 17: 1095-100. PMID: 4608227
- Antimicrobial activity of juncusol, a novel 9-10-dihydrophenanthrene from the marsh plant Juncus roemerianus. | Chapatwala, KD., et al. 1981. Life Sci. 29: 1997-2001. PMID: 6796796
- Regio- and stereospecific oxidation of 9,10-dihydroanthracene and 9,10-dihydrophenanthrene by naphthalene dioxygenase: structure and absolute stereochemistry of metabolites. | Resnick, SM. and Gibson, DT. 1996. Appl Environ Microbiol. 62: 3355-9. PMID: 8795226
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
9,10-Dihydrophenanthrene, 5 g | sc-254920 | 5 g | $106.00 |
