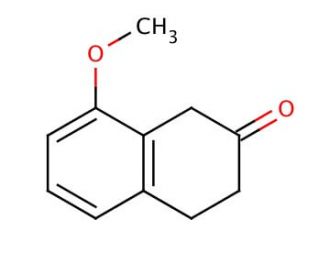
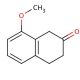
Molecular structure of 8-Methoxy-2-tetralone, CAS Number: 5309-19-3
8-Methoxy-2-tetralone (CAS 5309-19-3)
CAS Number:
5309-19-3
Molecular Weight:
176.21
Molecular Formula:
C11H12O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
8-Methoxy-2-tetralone is slightly soluble in water but highly soluble in organic solutions like ethanol, acetone, and chloroform. It′s used as a standard reference substance for identifying and determining its concentration in different samples. In vitro it is observed to enhance the function of antioxidant enzymes. It′s postulated that 8-Methoxy-2-tetralone acts by influencing various cellular signaling processes, particularly the nuclear factor kappa-B and mitogen-activated protein kinase pathways.
8-Methoxy-2-tetralone (CAS 5309-19-3) References
- Asymmetric reduction of substituted 2‐tetralones by Thermoanaerobacter pseudoethanolicus secondary alcohol dehydrogenase | Bsharat, O., Musa, M. M., Vieille, C., Oladepo, S. A., Takahashi, M., & Hamdan, S. M. ChemCatChem. 9(8): 1487-1493.
- Novel transformation of methoxy tetralones during demethylation with boron trifluoride etherate and acetic anhydride | Banerjee, A. K., Bedoya, L., Vera, W. J., Melean, C., Mora, H., Laya, M. S., & Alonso, M. Synthetic communications. 34(18): 3399-3408.
- Synthesis and characterization of monohydroxylated derivatives of 7H-dibenzo[c,g]carbazole. | Xue, WL. and Warshawsky, D. 1992. Chem Res Toxicol. 5: 130-3. PMID: 1581529
- 2-(Alkylamino)tetralin derivatives: interaction with 5-HT1A serotonin binding sites. | Naiman, N., et al. 1989. J Med Chem. 32: 253-6. PMID: 2521252
- Irreversible blockade of central 5-HT1A receptor binding sites by the photoaffinity probe 8-methoxy-3'-NAP-amino-PAT. | Emerit, MB., et al. 1986. Eur J Pharmacol. 127: 67-81. PMID: 2944752
- Identification of presynaptic serotonin autoreceptors using a new ligand: 3H-PAT. | Gozlan, H., et al. Nature. 305: 140-2. PMID: 6225026
- 8-Hydroxy-2-(alkylamino)tetralins and related compounds as central 5-hydroxytryptamine receptor agonists. | Arvidsson, LE., et al. 1984. J Med Chem. 27: 45-51. PMID: 6418888
- Synthesis of (+)-(R)- and (-)-(S)-trans-8-hydroxy-2-[N-n-propyl-N-(3'-iodo-2'-propenyl)] aminotetralin: new 5-HT1A receptor ligands. | Zhuang, ZP., et al. 1995. Chirality. 7: 452-8. PMID: 7577352
- Centrally acting serotonergic and dopaminergic agents. 2. Synthesis and structure-activity relationships of 2,3,3a,4,9,9a-hexahydro-1H-benz[f]indole derivatives. | Lin, CH., et al. 1993. J Med Chem. 36: 1069-83. PMID: 8097538
- 2-Amido-8-methoxytetralins: a series of nonindolic melatonin-like agents. | Copinga, S., et al. 1993. J Med Chem. 36: 2891-8. PMID: 8411005
- Centrally acting serotonergic agents. Synthesis and structure-activity relationships of C-1- or C-3-substituted derivatives of 8-hydroxy-2-(di-n-propylamino)tetralin. | Lin, CH., et al. 1993. J Med Chem. 36: 671-82. PMID: 8459396
- Asymmetric reduction of methoxy substituted β-tetralones using transfer hydrogenation | Mogi, M., Fuji, K., & Node, M. 2004. Tetrahedron: Asymmetry. 15(23): 3715-3717.
- Stereoselective Transaminase‐Mediated Synthesis of Serotonin and Melatonin Receptor Agonists | Baud, D., Tappertzhofen, N., Moody, T. S., Ward, J. M., & Hailes, H. C. 2022. Advanced Synthesis & Catalysis. 364(9): 1564-1572.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
8-Methoxy-2-tetralone, 1 g | sc-257018 | 1 g | $77.00 |
