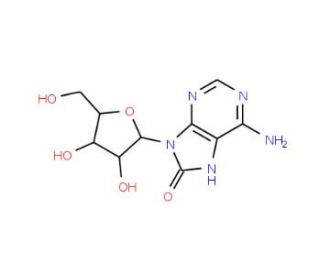
8-Hydroxyadenosine (CAS 29851-57-8)
QUICK LINKS
8-Hydroxyadenosine is a modified nucleoside in which an hydroxyl group (-OH) is added to the adenine base at the 8th position. In biochemical contexts, modifications like the one seen in 8-hydroxyadenosine are of interest for their potential roles in regulating nucleic acid function, stability, and interactions. Such modifications can occur naturally and are part of the cellular processes that involve RNA metabolism, potentially affecting processes like transcription, translation, and the regulation of gene expression. The presence of modified nucleosides can also impact the structure and function of RNA molecules, influencing their role in catalysis, structure, and the regulation of cellular processes. Additionally, the study of modified nucleosides like 8-hydroxyadenosine contributes to our understanding of cellular response mechanisms to stress and damage, such as oxidative stress, which can lead to the formation of oxidized nucleosides.
8-Hydroxyadenosine (CAS 29851-57-8) References
- Redox ribonucleosides. Isolation and characterization of 5-hydroxyuridine, 8-hydroxyguanosine, and 8-hydroxyadenosine from Torula yeast RNA. | Yanagawa, H., et al. 1992. J Biol Chem. 267: 13320-6. PMID: 1618833
- Structure of oxidatively damaged nucleic acid adducts. 3. Tautomerism, ionization and protonation of 8-hydroxyadenosine studied by 15N NMR spectroscopy. | Cho, BP. and Evans, FE. 1991. Nucleic Acids Res. 19: 1041-7. PMID: 1850508
- Purine 8-substitution modulates the ribonuclease L binding and activation abilities of 2',5'-oligoadenylates. | Kanou, M., et al. 1991. Biochem Biophys Res Commun. 176: 769-74. PMID: 2025289
- Isolation and characterization of phosmidosine. A new antifungal nucleotide antibiotic. | Uramoto, M., et al. 1991. J Antibiot (Tokyo). 44: 375-81. PMID: 2032945
- Chemical synthesis and biological activities of analogues of 2',5'-oligoadenylates containing 8-substituted adenosine derivatives. | Kanou, M., et al. 1990. Nucleic Acids Res. 18: 4439-46. PMID: 2167468
- Oxygen dependence of product formation in irradiated adenosine 5'-monophosphate. | Fuciarelli, AF., et al. 1988. Radiat Res. 113: 447-57. PMID: 3347702
- Characterization and biological activity of 8-substituted analogues of 2',5'-oligoadenylates. | Nagai, K., et al. 1993. Biochim Biophys Acta. 1156: 321-6. PMID: 8461324
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
8-Hydroxyadenosine, 50 mg | sc-284840 | 50 mg | $235.00 | |||
8-Hydroxyadenosine, 100 mg | sc-284840A | 100 mg | $337.00 |
