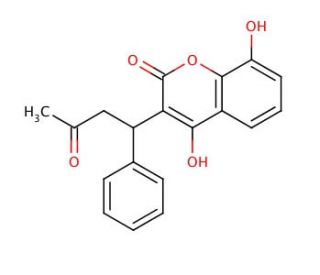
8-Hydroxy Warfarin (CAS 17834-04-7)
QUICK LINKS
8-Hydroxy Warfarin is a metabolite of warfarin that is researched mainly for its implications in metabolic pathways and environmental studies. In metabolic research, it serves as a marker to understand the biotransformation of warfarin and provides insight into the enzymes involved in oxidative metabolic processes. The study of 8-Hydroxy Warfarin is essential for elucidating the metabolic fate of substances within organisms and for understanding the kinetics of metabolite formation and degradation. In environmental contexts, it is used to track the presence and persistence of warfarin in ecosystems, helping to assess the impact of this compound on wildlife and environmental health. Additionally, 8-Hydroxy Warfarin′s properties may be investigated in the synthesis of new compounds, where knowledge of its chemical behavior can inform the design of molecules with desired characteristics.
8-Hydroxy Warfarin (CAS 17834-04-7) References
- The rapid identification of a new metabolite of warfarin via a chemical ionization mass spectrometry ion doublet technique. | Pohl, LR., et al. 1975. Biomed Mass Spectrom. 2: 23-30. PMID: 1131390
- Quantitative liquid chromatography/mass spectrometry/mass spectrometry warfarin assay for in vitro cytochrome P450 studies. | Zhang, ZY., et al. 2001. Anal Biochem. 298: 40-9. PMID: 11673893
- Polymorphism induced sensitivity to warfarin: a review of the literature. | Palkimas, MP., et al. 2003. J Thromb Thrombolysis. 15: 205-12. PMID: 14739630
- Comparison of the rat liver microsomal metabolism of the enantiomers of warfarin and 4'-nitrowarfarin (acenocoumarol). | Hermans, JJ. and Thijssen, HH. 1991. Xenobiotica. 21: 295-307. PMID: 1862655
- Stereoselectivity in pharmacokinetics: a general theory. | Levy, RH. and Boddy, AV. 1991. Pharm Res. 8: 551-6. PMID: 1866367
- Warfarin is an effective modifier of multiple UDP-glucuronosyltransferase enzymes: evaluation of its potential to alter the pharmacokinetics of zidovudine. | Sun, H., et al. 2015. J Pharm Sci. 104: 244-56. PMID: 25393417
- Comparison of enzyme kinetics of warfarin analyzed by LC-MS/MS QTrap and differential mobility spectrometry. | Shaik, AN., et al. 2016. J Chromatogr B Analyt Technol Biomed Life Sci. 1008: 164-173. PMID: 26655108
- Aspects of anticoagulant action: a review of the pharmacology, metabolism and toxicology of warfarin and congeners. | Sutcliffe, FA., et al. 1987. Rev Drug Metab Drug Interact. 5: 225-72. PMID: 3333881
- Interaction of the anticoagulant drug warfarin and its metabolites with human plasma albumin. | O'Reilly, RA. 1969. J Clin Invest. 48: 193-202. PMID: 5765021
- Composition of cytochrome P-450 isozymes from hepatic microsomes of C57BL/6 and DBA/2 mice assessed by warfarin metabolism, immunoinhibition, and immunoelectrophoresis with anti-(rat cytochrome P-450). | Kaminsky, LS., et al. 1984. Eur J Biochem. 141: 141-8. PMID: 6723656
- Biochemical applications of a quantitative high-pressure liquid chromatographic assay of warfarin and its metabolites. | Fasco, MJ., et al. 1977. J Chromatogr. 131: 365-73. PMID: 853101
- Human cytochromes P4501A1 and P4501A2: R-warfarin metabolism as a probe. | Zhang, Z., et al. 1995. Drug Metab Dispos. 23: 1339-46. PMID: 8689941
- Characterization of purified human recombinant cytochrome P4501A1-Ile462 and -Val462: assessment of a role for the rare allele in carcinogenesis. | Zhang, ZY., et al. 1996. Cancer Res. 56: 3926-33. PMID: 8752159
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
8-Hydroxy Warfarin, 5 mg | sc-210688 | 5 mg | $520.00 |
