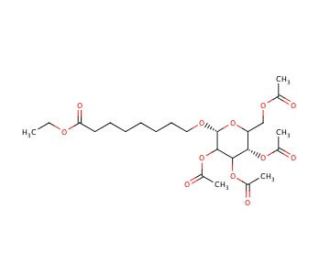
8-Ethoxycarbonyloctanoyl-2′,3′,4′,6-tetra-O-acetyl-β-D-galactopyranoside
QUICK LINKS
8-Ethoxycarbonyloctanoyl-2′,3′,4′,6-tetra-O-acetyl-β-D-galactopyranoside, often abbreviated as ECOATG, serves as a valuable tool in glycoscience research, particularly in the synthesis of glycolipids. Its mechanism of action involves its function as an acyl donor in glycosylation reactions, where it transfers the octanoyl group to the hydroxyl group of the galactopyranoside moiety. This chemical is commonly used in the synthesis of glycolipid derivatives, allowing researchers to study their structural features, physicochemical properties, and biological activities. ECOATG has been employed in the preparation of glycolipid-based biomaterials for applications in drug delivery, tissue engineering, and immunotherapy. Furthermore, it has found utility in the development of glycosylation methodologies, including enzymatic and chemical approaches, for the efficient synthesis of complex carbohydrates. Its versatility, ease of handling, and compatibility with various synthetic strategies make it a valuable reagent in the field of glycoscience, enabling the synthesis of diverse glycolipid analogs and derivatives for fundamental research and applied purposes.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
8-Ethoxycarbonyloctanoyl-2',3',4',6-tetra-O-acetyl-β-D-galactopyranoside, 250 mg | sc-221130 | 250 mg | $300.00 |
