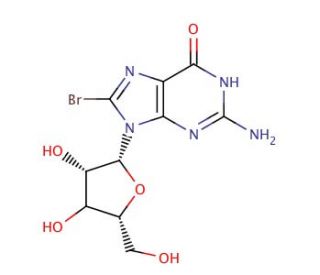
8-Bromoguanosine (CAS 4016-63-1)
QUICK LINKS
8-Bromoguanosine (8-Br-G) is an analogue of guanosine, a vital purine nucleoside found universally in organisms. Researchers have harnessed the versatility of 8-Bromoguanosine to investigate how modified nucleosides influence DNA replication, gene expression, and protein-nucleic acid interactions. Furthermore, it has been instrumental in unveiling the effects of modified nucleosides on the activity, structure, and function of enzymes involved in DNA replication and transcription. Although the exact mechanism remains unclear, it is postulated that 8-Bromoguanosine binds to specific proteins, disrupting their normal function and subsequently influencing the activity and structure of enzymes or proteins involved in DNA replication and transcription.
8-Bromoguanosine (CAS 4016-63-1) References
- Restricting the conformational heterogeneity of RNA by specific incorporation of 8-bromoguanosine. | Proctor, DJ., et al. 2003. J Am Chem Soc. 125: 2390-1. PMID: 12603116
- Efficacy of 8-bromoguanosine against murine cutaneous leishmaniasis induced with Leishmania amazonensis. | Barão, SC. and Giorgio, S. 2003. Chemotherapy. 49: 159-62. PMID: 12886049
- Synthesis of nucleoside libraries on solid support. II. 2,6,8-Trisubstituted purine nucleosides using 8-bromoguanosine as precursor. | Koh, YH., et al. 2004. Nucleosides Nucleotides Nucleic Acids. 23: 501-7. PMID: 15043170
- Folding thermodynamics and kinetics of YNMG RNA hairpins: specific incorporation of 8-bromoguanosine leads to stabilization by enhancement of the folding rate. | Proctor, DJ., et al. 2004. Biochemistry. 43: 14004-14. PMID: 15518549
- Structure of a human telomeric DNA sequence stabilized by 8-bromoguanosine substitutions, as determined by NMR in a K+ solution. | Matsugami, A., et al. 2007. FEBS J. 274: 3545-3556. PMID: 17561958
- Formation of 8-S-L-cysteinylguanosine from 8-bromoguanosine and cysteine. | Suzuki, T., et al. 2013. Bioorg Med Chem Lett. 23: 3864-7. PMID: 23714713
- Active transformation of tolerogenic to immunogenic signals in T and B cells by 8-bromoguanosine. | Scheuer, WV., et al. 1985. J Immunol. 135: 2962-6. PMID: 2413105
- Structural characterization of a dimer of RNA duplexes composed of 8-bromoguanosine modified CGG trinucleotide repeats: a novel architecture of RNA quadruplexes. | Gudanis, D., et al. 2016. Nucleic Acids Res. 44: 2409-16. PMID: 26743003
- Antiparallel RNA G-quadruplex Formed by Human Telomere RNA Containing 8-Bromoguanosine. | Xiao, CD., et al. 2017. Sci Rep. 7: 6695. PMID: 28751647
- Synthesis and surface characterization of new triplex polymer of Ag(I) and mixture nucleosides: cytidine and 8-bromoguanosine. | Al-Mahamad, LLG. 2019. Heliyon. 5: e01609. PMID: 31193246
- Singlet O2 Reactions with Radical Cations of 8-Bromoguanine and 8-Bromoguanosine: Guided-Ion Beam Mass Spectrometric Measurements and Theoretical Treatments. | Benny, J., et al. 2022. J Phys Chem A. 126: 68-79. PMID: 34941276
- Large, activated B cells are the primary B-cell target of 8-bromoguanosine and 8-mercaptoguanosine. | Wicker, LS., et al. 1987. Cell Immunol. 106: 318-29. PMID: 3494541
- Crystal and molecular structure of 8-bromoguanosine and 8-bromoadenosine, two purine nucleosides in the syn conformation. | Tavale, SS. and Sobell, HM. 1970. J Mol Biol. 48: 109-23. PMID: 5448585
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
8-Bromoguanosine, 5 g | sc-210669 | 5 g | $176.00 |
