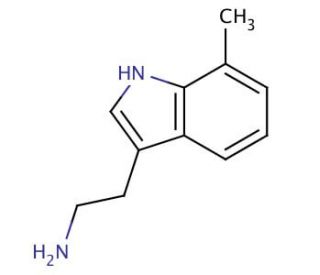
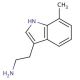
7-Methyltryptamine (CAS 14490-05-2)
QUICK LINKS
7-Methyltryptamine is a compound that functions as a serotonin receptor agonist, targeting the 5-HT1 and 5-HT2 receptor subtypes. It acts by binding to these receptors and activating intracellular signaling pathways, leading to a cascade of cellular responses. 7-Methyltryptamine modulates neurotransmitter release and neuronal activity in the central nervous system, influencing various physiological processes. At the molecular level, 7-Methyltryptamine interacts with specific binding sites on serotonin receptors, altering their conformation and initiating downstream signaling events. 7-Methyltryptamine′s mechanism of action involves the modulation of serotonin receptor activity, which can impact synaptic transmission and neuronal excitability. In a development setting, 7-Methyltryptamine is utilized to investigate the specific effects of serotonin receptor activation on cellular and neuronal function, providing insights into the role of these receptors in various physiological and pathological conditions.
7-Methyltryptamine (CAS 14490-05-2) References
- Interactions of tryptamine derivatives with serotonin transporter species variants implicate transmembrane domain I in substrate recognition. | Adkins, EM., et al. 2001. Mol Pharmacol. 59: 514-23. PMID: 11179447
- Serotonergic and dopaminergic influence of the duration of embryogenesis and intracapsular locomotion of Lymnaea stagnalis L. | Filla, A., et al. 2004. Acta Biol Hung. 55: 315-21. PMID: 15270248
- Voltage-dependent inhibition of recombinant NMDA receptor-mediated currents by 5-hydroxytryptamine. | Kloda, A. and Adams, DJ. 2005. Br J Pharmacol. 144: 323-30. PMID: 15655527
- Enantioselective Brønsted acid-catalyzed N-acyliminium cyclization cascades. | Muratore, ME., et al. 2009. J Am Chem Soc. 131: 10796-7. PMID: 19606900
- On the transmitter at the A-to-B cell in Aplysia californica. | Gaillard, WD. and Carpenter, DO. 1986. Brain Res. 373: 311-5. PMID: 2424554
- Alpha-ethyltryptamines as dual dopamine-serotonin releasers. | Blough, BE., et al. 2014. Bioorg Med Chem Lett. 24: 4754-4758. PMID: 25193229
- 5-Hydroxytryptamine stimulates inositol phosphate production in a cell-free system from blowfly salivary glands. Evidence for a role of GTP in coupling receptor activation to phosphoinositide breakdown. | Litosch, I., et al. 1985. J Biol Chem. 260: 5464-71. PMID: 2985595
- Ionic mechanisms and receptor properties underlying the responses of molluscan neurones to 5-hydroxytryptamine. | Gerschenfeld, HM. and Paupardin-Tritsch, D. 1974. J Physiol. 243: 427-56. PMID: 4155767
- On the transmitter function of 5-hydroxytryptamine at excitatory and inhibitory monosynaptic junctions. | Gerschenfeld, HM. and Paupardin-Tritsch, D. 1974. J Physiol. 243: 457-81. PMID: 4155768
- Purification and properties of strictosidine synthase, the key enzyme in indole alkaloid formation. | Treimer, JF. and Zenk, MH. 1979. Eur J Biochem. 101: 225-33. PMID: 510306
- Presynaptic modulation of synaptic transmission in Helix pomatia: the effects of serotonin and dopamine antagonists. | Juel, C. 1981. Neuropharmacology. 20: 323-6. PMID: 6117031
- [Significance of serotonin in the activity of the taste receptor apparatus of the frog Rana temporaria]. | Esakov, AI., et al. 1983. Zh Evol Biokhim Fiziol. 19: 62-7. PMID: 6601342
- Modulation of swimming in Tritonia: excitatory and inhibitory effects of serotonin. | McClellan, AD., et al. 1994. J Comp Physiol A. 174: 257-66. PMID: 7908336
- Acid-catalyzed deuterium exchange of the indole ring protons in tryptamine derivatives. | Kang, S., et al. 1977. J Org Chem. 42: 3769-72. PMID: 915589
- Efflux studies allow further characterisation of the noradrenaline and 5-hydroxytryptamine transporters in rat lungs. | James, KM. and Bryan-Lluka, LJ. 1997. Naunyn Schmiedebergs Arch Pharmacol. 356: 126-33. PMID: 9228199
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
7-Methyltryptamine, 100 mg | sc-227121 | 100 mg | $260.00 |
