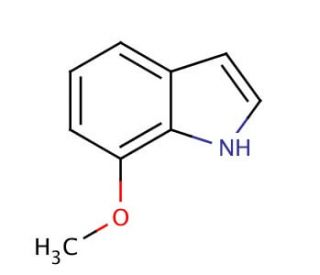
7-Methoxyindole (CAS 3189-22-8)
QUICK LINKS
7-methoxyindole, is indole derivative, a small molecule that can be synthesized in the laboratory, has found utility in diverse scientific fields for research purposes. Scientists have delved into the potential roles of 7-methoxyindole in various scientific research applications. It has served as a model compound to investigate the impacts of indole derivatives. Furthermore, it has been instrumental in studying the effects of indole-3-carboxaldehyde on the synthesis of indole derivatives. Additionally, 7-methoxyindole has shed light on the influence of indole derivatives on enzyme activity, notably the cytochrome P450 enzymes. While the precise mechanism of action of 7-methoxyindole remains elusive, it is known to interact with cytochrome P450 enzymes.
7-Methoxyindole (CAS 3189-22-8) References
- Microbial transformation of aspidospermine. | Lin, SK., et al. 1975. J Pharm Sci. 64: 2021-2. PMID: 1206502
- Utilization of indole analogs by carrot and tobacco cell tryptophan synthase in vivo and in vitro. | Widholm, JM. 1981. Plant Physiol. 67: 1101-4. PMID: 16661817
- Direct palladium-catalyzed C-3 arylation of free (NH)-indoles with aryl bromides under ligandless conditions. | Bellina, F., et al. 2008. J Org Chem. 73: 5529-35. PMID: 18543968
- Design, synthesis, and biological evaluation of aminoalkylindole derivatives as cannabinoid receptor ligands with potential for treatment of alcohol abuse. | Vasiljevik, T., et al. 2013. J Med Chem. 56: 4537-50. PMID: 23631463
- Biomimetic Enantioselective Total Synthesis of (-)-Mycoleptodiscin A. | Dethe, DH., et al. 2016. Org Lett. 18: 6392-6395. PMID: 27978704
- Methylindoles and Methoxyindoles are Agonists and Antagonists of Human Aryl Hydrocarbon Receptor. | Stepankova, M., et al. 2018. Mol Pharmacol. 93: 631-644. PMID: 29626056
- Design, Synthesis and Evaluation of New Indolylpyrimidylpiperazines for Gastrointestinal Cancer Therapy. | Tan, A., et al. 2019. Molecules. 24: PMID: 31614517
- Antibiofilm and Antivirulence Properties of Indoles Against Serratia marcescens. | Sethupathy, S., et al. 2020. Front Microbiol. 11: 584812. PMID: 33193228
- The Anticancer Agent 3,3'-Diindolylmethane Inhibits Multispecies Biofilm Formation by Acne-Causing Bacteria and Candida albicans. | Kim, YG., et al. 2022. Microbiol Spectr. 10: e0205621. PMID: 35107361
- Free energy perturbation (FEP)-guided scaffold hopping. | Wu, D., et al. 2022. Acta Pharm Sin B. 12: 1351-1362. PMID: 35530128
- Characterization of marine-derived halogenated indoles as ligands of the aryl hydrocarbon receptor. | King, J., et al. 2022. Toxicol Rep. 9: 1198-1203. PMID: 36518459
- The Impact of Indoles Activating the Aryl Hydrocarbon Receptor on Androgen Receptor Activity in the 22Rv1 Prostate Cancer Cell Line. | Zgarbová, E. and Vrzal, R. 2022. Int J Mol Sci. 24: PMID: 36613955
- Molecular orbital calculations of magnetic circular dichroism spectra: a benzene-indole sequence. | Miles, DW. and Eyring, H. 1973. Proc Natl Acad Sci U S A. 70: 3754-8. PMID: 4521200
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
7-Methoxyindole, 1 g | sc-254908 | 1 g | $110.00 |
