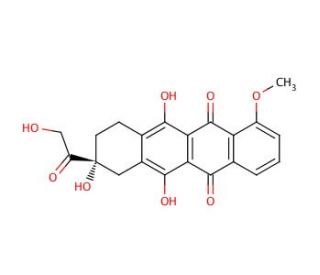
7-Deoxy Doxorubicin Aglycone (CAS 38554-25-5)
See product citations (1)
QUICK LINKS
7-Deoxy Doxorubicin Aglycone, a derivative of the anthracycline antibiotic doxorubicin, is a compound of significant interest in biochemical research, particularly in the study of cancer cell biology. Unlike its parent compound, doxorubicin, which is widely recognized for its clinical applications, 7-Deoxy Doxorubicin Aglycone lacks the sugar moiety, altering its mechanism of action and its interaction with cellular targets. This structural modification results in a compound with distinct physicochemical properties and biological activities, making it useful for investigating the nuances of anthracycline action at the molecular level. Its role in research often focuses on understanding the pathways of cellular apoptosis, mechanisms of drug resistance, and the dynamics of DNA intercalation and topoisomerase II inhibition. By studying 7-Deoxy Doxorubicin Aglycone, researchers aim to elucidate the fundamental processes that underpin anthracycline-induced cytotoxicity, offering insights into the development of novel anticancer strategies that circumvent the limitations and side effects associated with traditional chemotherapy agents.
7-Deoxy Doxorubicin Aglycone (CAS 38554-25-5) References
- Anthracyclines: recent developments in their separation and quantitation. | Zagotto, G., et al. 2001. J Chromatogr B Biomed Sci Appl. 764: 161-71. PMID: 11817026
- Bepridil in combination with anthracyclines to reverse anthracycline resistance in cancer patients. | van Kalken, CK., et al. 1991. Eur J Cancer. 27: 739-44. PMID: 1829915
- The Role of the Metabolism of Anticancer Drugs in Their Induced-Cardiotoxicity. | Reis-Mendes, AF., et al. 2015. Curr Drug Metab. 17: 75-90. PMID: 26526839
- Adsorption of doxorubicin on citrate-capped gold nanoparticles: insights into engineering potent chemotherapeutic delivery systems. | Curry, D., et al. 2015. Nanoscale. 7: 19611-9. PMID: 26549208
- Molecular Dynamics Simulations of the pH-Dependent Adsorption of Doxorubicin on Carbon Quantum Dots. | Wolski, P. 2021. Mol Pharm. 18: 257-266. PMID: 33325232
- Metabolism of epidoxorubicin in animals: absence of glucuronidation. | Maessen, PA., et al. 1987. Cancer Chemother Pharmacol. 20: 85-7. PMID: 3476219
- Semiquinone free radical formation by daunorubicin aglycone incorporated into the cellular membranes of intact Chinese hamster ovary cells. | Malisza, KL., et al. 1996. Free Radic Res. 24: 9-18. PMID: 8747888
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
7-Deoxy Doxorubicin Aglycone, 1 mg | sc-210604 | 1 mg | $463.00 | |||
7-Deoxy Doxorubicin Aglycone, 10 mg | sc-210604A | 10 mg | $3269.00 |
