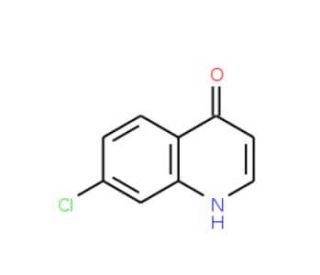
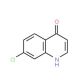
7-Chloro-4-hydroxyquinoline (CAS 86-99-7)
QUICK LINKS
7-Chloro-4-hydroxyquinoline possesses versatile synthetic pathways, being obtainable from either 4-hydroxyquinoline or 4-chloroquinoline. In the synthesis from 4-hydroxyquinoline, the reaction involves chlorine gas reacting with the compound in the presence of potassium hydroxide as a base. This reaction results in the production of 7-Chloro-4-hydroxyquinoline as well as potassium chloride. Conversely, the synthesis from 4-chloroquinoline entails the reaction of the compound with hydrogen peroxide in the presence of sodium hydroxide as a base, yielding 7-Chloro-4-hydroxyquinoline along with sodium chloride as the primary products. The applications of 7-Chloro-4-hydroxyquinoline in scientific research encompass a broad range. It has found utility as a catalyst in organic synthesis, an enzyme inhibitor, and a fluorescent probe in imaging studies. While the exact mechanism of action of 7-Chloro-4-hydroxyquinoline remains not fully elucidated, it is believed to act as an enzyme inhibitor by binding to the active site of the enzyme, thereby impeding its activity. Furthermore, 7-Chloro-4-hydroxyquinoline has demonstrated interactions with DNA and RNA, potentially influencing gene expression.e
7-Chloro-4-hydroxyquinoline (CAS 86-99-7) References
- Antioxidative or prooxidative effect of 4-hydroxyquinoline derivatives on free-radical-initiated hemolysis of erythrocytes is due to its distributive status. | Liu, ZQ., et al. 2002. Biochim Biophys Acta. 1570: 97-103. PMID: 11985893
- Computational Studies of Hydroxychloroquine and Chloroquine Metabolites as Possible Candidates for Coronavirus (COVID-19) Treatment. | Vaidya, NA. and Vyas, R. 2020. Front Pharmacol. 11: 569665. PMID: 33364944
- Nopol-Based Quinoline Derivatives as Antiplasmodial Agents. | Nyamwihura, RJ., et al. 2021. Molecules. 26: PMID: 33673007
- An Update on Pharmacological Relevance and Chemical Synthesis of Natural Products and Derivatives with Anti SARS-CoV-2 Activity. | Shagufta,. and Ahmad, I. 2021. ChemistrySelect. 6: 11502-11527. PMID: 34909460
- Chloroquine metabolism in man: urinary excretion of 7-chloro-4-hydroxyquinoline and 7-chloro-4-aminoquinoline metabolites. | Brown, ND., et al. 1985. J Chromatogr. 345: 209-14. PMID: 3841350
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
7-Chloro-4-hydroxyquinoline, 25 g | sc-227116 | 25 g | $30.00 |
