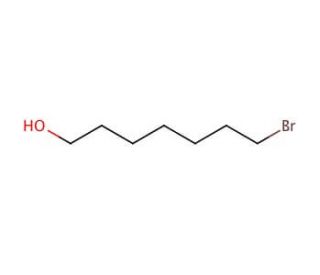
7-Bromo-1-heptanol (CAS 10160-24-4)
QUICK LINKS
7-Bromo-1-heptanol has garnered attention in scientific research due to its unique chemical properties and diverse applications. One prominent area of study involves its role as a versatile building block in organic synthesis, particularly in the preparation of complex organic molecules and polymers. Researchers have exploited the bromo functional group′s reactivity in various chemical transformations, including nucleophilic substitutions and cross-coupling reactions, to facilitate the synthesis of structurally diverse compounds. Moreover, 7-Bromo-1-heptanol has been investigated for its potential as a starting material in the synthesis of surfactants and specialty chemicals, owing to its amphiphilic nature and ability to modify surface properties. Additionally, its utilization as a precursor in the fabrication of functionalized materials, such as polymer brushes and self-assembled monolayers, has been explored for applications in surface modification and bioconjugation chemistry. Furthermore, studies have explaind its mechanistic pathways in organic reactions, contributing to the development of efficient synthetic methodologies. Overall, 7-Bromo-1-heptanol serves as a valuable tool in organic synthesis and materials science research, offering opportunities for the creation of novel compounds and materials with tailored properties for various scientific applications.
7-Bromo-1-heptanol (CAS 10160-24-4) References
- Synthesis of 'Porphyrin-linker-Thiol' molecules with diverse linkers for studies of molecular-based information storage. | Gryko, DT., et al. 2000. J Org Chem. 65: 7345-55. PMID: 11076590
- Synthesis and bioactivity of linear oligomers related to polymeric alkylpyridinium metabolites from the Mediterranean sponge Reniera sarai. | Mancini, I., et al. 2004. Org Biomol Chem. 2: 1368-75. PMID: 15105928
- The introduction of pi-pi stacking moieties for fabricating stable micellar structure: formation and dynamics of disklike micelles. | Song, B., et al. 2005. Angew Chem Int Ed Engl. 44: 4731-5. PMID: 15995994
- First total syntheses of (Z)-15-methyl-10-hexadecenoic acid and the (Z)-13-methyl-8-tetradecenoic acid. | Carballeira, NM., et al. 2007. Chem Phys Lipids. 145: 37-44. PMID: 17125759
- Discovery of a Small-Molecule Degrader of Bromodomain and Extra-Terminal (BET) Proteins with Picomolar Cellular Potencies and Capable of Achieving Tumor Regression. | Zhou, B., et al. 2018. J Med Chem. 61: 462-481. PMID: 28339196
- Quantitative and time-resolved monitoring of organelle and protein delivery to the lysosome with a tandem fluorescent Halo-GFP reporter. | Rudinskiy, M., et al. 2022. Mol Biol Cell. 33: ar57. PMID: 35108065
- Mitochondrial sodium/calcium exchanger (NCLX) regulates basal and starvation-induced autophagy through calcium signaling. | Ramos, VM., et al. 2024. FASEB J. 38: e23454. PMID: 38315457
- Urinary excretion and metabolism of the α-pyrrolidinophenone designer drug 1-phenyl-2-(pyrrolidin-1-yl)octan-1-one (PV9) in humans | Noriaki Shima, Hidenao Kakehashi, Shuntaro Matsuta, Hiroe Kamata, Shihoko Nakano, Keiko Sasaki, Tooru Kamata, Hiroshi Nishioka, Kei Zaitsu, Takako Sato, Akihiro Miki, Munehiro Katagi & Hitoshi Tsuchihashi. 2015. Forensic Toxicology. 33: 279–294.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
7-Bromo-1-heptanol, 1 g | sc-223734 | 1 g | $83.00 | |||
7-Bromo-1-heptanol, 5 g | sc-223734A | 5 g | $294.00 |
