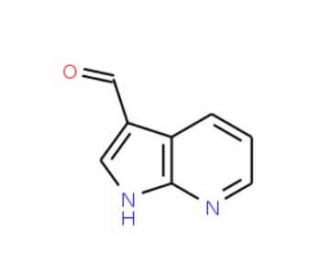
Molecular structure of 7-Azaindole-3-carboxaldehyde, CAS Number: 4649-09-6
7-Azaindole-3-carboxaldehyde (CAS 4649-09-6)
Alternate Names:
1H-Pyrrolo[2,3-b]pyridine-3-carbaldehyde
Application:
7-Azaindole-3-carboxaldehyde is a useful starter in 7-azaindole chemistry
CAS Number:
4649-09-6
Purity:
≥96%
Molecular Weight:
146.15
Molecular Formula:
C8H6N2O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
7-Azaindole-3-carboxaldehyde is a useful starter in 7-azaindole chemistry.
7-Azaindole-3-carboxaldehyde (CAS 4649-09-6) References
- Synthesis and evaluation of fluorine-substituted 1H-pyrrolo[2,3-b]pyridine derivatives for dopamine D4 receptor imaging. | Oh, SJ., et al. 2004. Bioorg Med Chem. 12: 5505-13. PMID: 15465327
- Synthesis of 1-p-chlorobenzyl-7-azaindole-3- -piperidylmethanol as a potential antimalarial agent. | Verbiscar, AJ. 1972. J Med Chem. 15: 149-52. PMID: 4400149
- Radiosynthesis of 3-{[4-(4-[18F]fluorobenzyl)]piperazin-1-yl}methyl-1H-pyrrolo[2,3-b]pyridine: A potential dopamine D4 receptor imaging agent | Hai-Bin, T., Duan-Zhi, Y., Jun-Ling, L., Lan, Z., Cun-Fu, Z., Yong-**an, W., & Wei, Z. 2003. Radiochimica Acta. 91(4): 241-245.
- Azaindole modified imine moiety as fluorescent probe for highly sensitive detection of Fe3+ ions | Kaur, K., Chaudhary, S., Singh, S., & Mehta, S. K. 2016. Sensors and Actuators B: Chemical. 232: 396-401.
- An azaindole–hydrazine imine moiety as sensitive dual cation chemosensor depending on surface plasmon resonance and emission properties | Kaur, K., Chaudhary, S., Singh, S., & Mehta, S. K. 2016. Sensors and Actuators B: Chemical. 222: 397-406.
- X-ray crystal structure, infrared, Raman and density functional studies of 7-azaindole-3-carboxaldehyde | Morzyk-Ociepa, B., Szmigiel, K., Turowska-Tyrk, I., & Michalska, D. 2017. Journal of Molecular Structure. 1128: 186-194.
- New mono-and dinuclear complexes of 7-azaindole-3-carboxaldehyde with palladium (II): Crystal structure, IR and Raman spectra, DFT calculations and in vitro antiproliferative activity | Morzyk-Ociepa, B., Szmigiel, K., Turowska-Tyrk, I., Malik-Gajewska, M., Banach, J., & Wietrzyk, J. 2018. Polyhedron. 153: 88-98.
- Platinum(II) and copper(II) complexes of 7-azaindole-3-carboxaldehyde: crystal structures, IR and Raman spectra, DFT calculations and in vitro antiproliferative activity of the platinum(II) complex | Morzyk-Ociepa, B., Szmigiel-Bakalarz, K., Nentwig, M., Oeckler, O., Malik-Gajewska, M., Turlej, E.,.. & Michalska, D. 2019. Inorganica Chimica Acta. 490: 68-77.
- 7-Azaindole-3-carboxylic acid and its Pt (II) and Pd (II) complexes: Crystal structure of the ligand, vibrational spectra, DFT calculations and in vitro antiproliferative activity | Szmigiel-Bakalarz, K., Nentwig, M., Oeckler, O., Malik-Gajewska, M., Filip-Psurska, B., & Morzyk-Ociepa, B. 2020. Journal of Molecular Structure. 1203: 127441.
- Synthesis, characterization and preliminary antibacterial evaluation against Staphylococcus aureus of a new 2, 4, 5-tri (hetero) arylimidazole derivative based on azaindole heterocycle | Ramos, N. L., Oliveira, R., Costa, S. P., & Raposo, M. M. M. 2021. Chemistry Proceedings. 8(1): 104.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
7-Azaindole-3-carboxaldehyde, 1 g | sc-254899 | 1 g | $54.00 |
