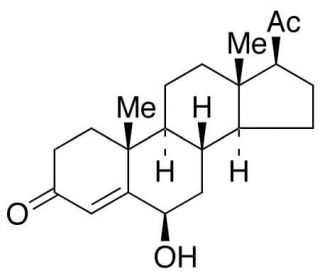
6β-Hydroxy Progesterone (CAS 604-19-3)
QUICK LINKS
6β-Hydroxy Progesterone, a metabolite of progesterone, showcases a unique profile in biochemical research due to its altered physiological activities when compared to its progenitor hormone. This compound is particularly significant in the study of steroid metabolism and hormone action mechanisms, offering insights into the intricate processes of enzymatic modification and the consequential modulation of hormone activity. Through its hydroxylation at the 6β position, it serves as a pivotal tool for elucidating the role of specific hydroxyl groups in the steroid nucleus on receptor binding affinity and activation, thus shedding light on the molecular underpinnings of steroid hormone action. Moreover, 6β-Hydroxy Progesterone is instrumental in the exploration of steroidogenic pathways, providing a window into the complexity of hormone synthesis and degradation, and the balance these processes maintain in physiological homeostasis. Its function and metabolism are critical in the context of endocrinology and biochemistry research, where understanding the nuances of hormone action can unveil new pathways for the regulation of biological systems.
6β-Hydroxy Progesterone (CAS 604-19-3) References
- Studies on Bacillus stearothermophilus. Part 1. Transformation of progesterone to a new metabolite 9,10-seco-4-pregnene-3,9,20-trione. | Al-Awadi, S., et al. 2001. J Steroid Biochem Mol Biol. 78: 493-8. PMID: 11738559
- Structural specificity of steroids in stimulating DNA synthesis and protooncogene expression in primary rat hepatocyte cultures. | Lee, CH. and Edwards, AM. 2002. J Steroid Biochem Mol Biol. 81: 25-36. PMID: 12127039
- Sodium ascorbate improves yield of urinary steroids during hydrolysis with Helix pomatia juice. | Christakoudi, S., et al. 2008. Steroids. 73: 309-19. PMID: 18177910
- Antioxidant activity of biotransformed sex hormones facilitated by Bacillus stearothermophilus. | Afzal, M., et al. 2010. Methods Mol Biol. 594: 349-56. PMID: 20072930
- 2β- and 16β-hydroxylase activity of CYP11A1 and direct stimulatory effect of estrogens on pregnenolone formation. | Mosa, A., et al. 2015. J Steroid Biochem Mol Biol. 150: 1-10. PMID: 25746800
- CYP106A2-A versatile biocatalyst with high potential for biotechnological production of selectively hydroxylated steroid and terpenoid compounds. | Schmitz, D., et al. 2018. Biochim Biophys Acta Proteins Proteom. 1866: 11-22. PMID: 28780179
- Celular and subcellular metabolism of progesterone by the human proliferative and secretory phase endometrium and myometrium. | Verma, U. and Laumas, KR. 1976. J Steroid Biochem. 7: 275-82. PMID: 933525
- Comparative effects of cytokines on constitutive and inducible expression of the gene encoding for the cytochrome P450 3A6 isoenzyme in cultured rabbit hepatocytes: consequences on progesterone 6beta-hydroxylation. | Calleja, C., et al. 1998. Biochem Pharmacol. 56: 1279-85. PMID: 9825726
- Progesterone 6-hydroxylation is catalysed by cytochrome P-450 in the moderate thermophile Bacillus thermoglucosidasius strain 12060. | Sideso, O., et al. 1998. J Steroid Biochem Mol Biol. 67: 163-9. PMID: 9877217
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
6β-Hydroxy Progesterone, 100 mg | sc-488157 | 100 mg | $396.00 |
