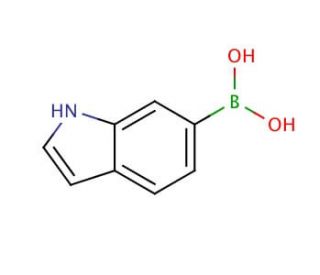
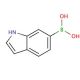
Molecular structure of 6-Indolylboronic acid, CAS Number: 147621-18-9
6-Indolylboronic acid (CAS 147621-18-9)
Alternate Names:
6-Indoleboronic acid; Indole-6-boronic acid
Application:
6-Indolylboronic acid is a chemical used in a study of racemization
CAS Number:
147621-18-9
Molecular Weight:
160.97
Molecular Formula:
C8H8BNO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
This is used in a study of racemization in Suzuki cross-couplings of arylamino acids. It is also used to as a reactnt in the synthesis of PI3K inhibitors, CHK1 inhibitors, and HIV-1 glycoprotein-41 fusion inhibitors. This product may contain different amounts of anhydride.
6-Indolylboronic acid (CAS 147621-18-9) References
- Racemization in suzuki couplings: a quantitative study using 4-hydroxyphenylglycine and tyrosine derivatives as probe molecules. | Prieto, M., et al. 2007. J Org Chem. 72: 1047-50. PMID: 17253834
- Microwave-Assisted Synthesis of a MK2 Inhibitor by Suzuki-Miyaura Coupling for Study in Werner Syndrome Cells. | Bagley, MC., et al. 2015. Pharmaceuticals (Basel). 8: 257-76. PMID: 26046488
- Enantioselective, Palladium-Catalyzed Conjugate Additions of Arylboronic Acids to Form Bis-benzylic Quaternary Stereocenters. | Kadam, AA., et al. 2017. Org Lett. 19: 4062-4065. PMID: 28737030
- Asymmetric cross-coupling of alkyl, alkenyl and (hetero)aryl nucleophiles with racemic allyl halides. | Schäfer, P., et al. 2017. Chem Commun (Camb). 53: 12499-12511. PMID: 29098227
- Discovery of a novel indole pharmacophore for the irreversible inhibition of myeloperoxidase (MPO). | Patnaik, A., et al. 2020. Bioorg Med Chem. 28: 115548. PMID: 32503688
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
6-Indolylboronic acid, 1 g | sc-239100 | 1 g | $84.00 |
