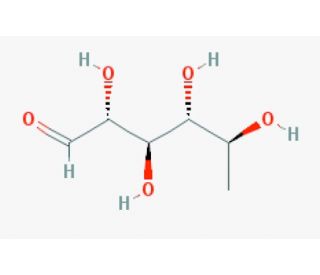
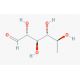
6-Deoxy-L-talose (CAS 7658-10-8)
QUICK LINKS
6-Deoxy-L-talose, a rare sugar and deoxyhexose, has captured scientific interest due to its unique stereochemistry and structural relationship to other monosaccharides. Its unusual lack of a hydroxyl group at the 6th carbon differentiates it from common hexoses, providing researchers with distinctive opportunities in carbohydrate chemistry and glycoscience. This compound is particularly valuable for exploring the role of deoxysugars in biological systems. Its structure is found in some bacterial polysaccharides, which has inspired studies on microbial glycan biosynthesis and metabolism. In research on microbial polysaccharides, 6-Deoxy-L-talose is studied to explain how bacteria synthesize and utilize such sugars in their glycoconjugates. By understanding these biosynthetic pathways, researchers can gain insights into bacterial virulence and identify potential targets for developing inhibitors against pathogenic strains. Moreover, 6-Deoxy-L-talose′s stereochemical specificity makes it useful as a chiral building block in synthetic chemistry, particularly in the design of complex carbohydrate derivatives. Investigations into the enzymatic synthesis and modification of this sugar also help clarify enzyme specificity and mechanism. Thus, 6-Deoxy-L-talose plays a crucial role in carbohydrate research, enabling the study of diverse biological processes and the development of new synthetic methodologies.
6-Deoxy-L-talose (CAS 7658-10-8) References
- Thymidine diphosphate-6-deoxy-L-lyxo-4-hexulose reductase synthesizing dTDP-6-deoxy-L-talose from Actinobacillus actinomycetemcomitans. | Nakano, Y., et al. 2000. J Biol Chem. 275: 6806-12. PMID: 10702238
- Structure of the O-polysaccharide of Aeromonas hydrophila O:34; a case of random O-acetylation of 6-deoxy-L-talose. | Knirel, YA., et al. 2002. Carbohydr Res. 337: 1381-6. PMID: 12204621
- 6-Deoxy-D-talan and 6-deoxy-L-talan. Novel serotype-specific polysaccharide antigens from Actinobacillus actinomycetemcomitans. | Shibuya, N., et al. 1991. J Biol Chem. 266: 16318-23. PMID: 1885566
- Novel process for producing 6-deoxy monosaccharides from l-fucose by coupling and sequential enzymatic method. | Shompoosang, S., et al. 2016. J Biosci Bioeng. 121: 1-6. PMID: 26031195
- 6-Deoxyhexoses from l-Rhamnose in the Search for Inducers of the Rhamnose Operon: Synergy of Chemistry and Biotechnology. | Liu, Z., et al. 2016. Chemistry. 22: 12557-65. PMID: 27439720
- Replacement of the L-iduronic acid unit of the anticoagulant pentasaccharide idraparinux by a 6-deoxy-L-talopyranose - Synthesis and conformational analysis. | Demeter, F., et al. 2018. Sci Rep. 8: 13736. PMID: 30213971
- Acinetobacter baumannii K106 and K112: Two Structurally and Genetically Related 6-Deoxy-l-talose-Containing Capsular Polysaccharides. | Kasimova, AA., et al. 2021. Int J Mol Sci. 22: PMID: 34073255
- The K26 capsular polysaccharide from Acinetobacter baumannii KZ-1098: Structure and cleavage by a specific phage depolymerase. | Kasimova, AA., et al. 2021. Int J Biol Macromol. 191: 182-191. PMID: 34537298
- Structure of the K87 capsular polysaccharide and KL87 gene cluster of Acinetobacter baumannii LUH5547 reveals a heptasaccharide repeating unit. | Arbatsky, NP., et al. 2021. Carbohydr Res. 509: 108439. PMID: 34555685
- Biological mechanisms involved in the formation of deoxy sugars. VII. Biosynthesis of 6-deoxy-L-talose. | Gaugler, RW. and Gabriel, O. 1973. J Biol Chem. 248: 6041-9. PMID: 4199258
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
6-Deoxy-L-talose, 5 mg | sc-217338 | 5 mg | $260.00 |
