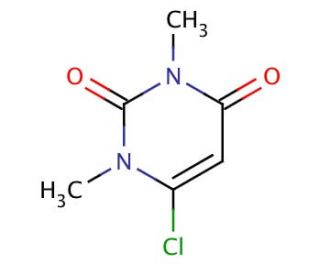
Molecular structure of 6-Chloro-1,3-dimethyluracil, CAS Number: 6972-27-6
6-Chloro-1,3-dimethyluracil (CAS 6972-27-6)
Alternate Names:
4-Chloro-1,3-dimethyluracil
CAS Number:
6972-27-6
Molecular Weight:
174.58
Molecular Formula:
C6H7ClN2O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
6-Chloro-1,3-dimethyluracil is a nucleobase constitute of nucleic acids. It acts as an intermediate in the synthesis of substituted uracils and derivatives. In vitro studies have demonstrated its cytotoxic effects. The synthesis of 6-Chloro-1,3-dimethyluracil can be achieved through the reaction of naphthalene with a nucleophilic nitrogen source, such as sodium carbonate, followed by chlorination with trifluoroacetic acid. Alternatively, it can be synthesized by reacting malonic acid with cyclen and uridine in the presence of copper metal. The reaction mechanism is thought to involve photoelectron transfer from the copper metal to the uridine ring.
6-Chloro-1,3-dimethyluracil (CAS 6972-27-6) References
- Acid-catalyzed photoreaction of 6-chloro-1,3-dimethyluracil and mesitylene: formation of photocycloadducts and their characterization. | Ohkura, K., et al. 2001. Chem Pharm Bull (Tokyo). 49: 384-90. PMID: 11310662
- Electrocyclic rearrangement of pentamethylcyclooctapyrimidine-2,4-diones: reaction pathway into a 9,11-diazapentacyclo-[6.4.0.0(1,3).0(2,5).0(4,8)]dodecane system and a 9,11-diazapentacyclo [6.4.0.0(1,3).0(2,6).0(4,8)]dodecane system. | Ohkura, K., et al. 2001. Photochem Photobiol. 74: 385-90. PMID: 11594050
- Electronic structure and biological activity of nucleobases. | Novak, I. and Kovac, B. 2005. Spectrochim Acta A Mol Biomol Spectrosc. 61: 2771-4. PMID: 16043077
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
6-Chloro-1,3-dimethyluracil, 1 g | sc-214368 | 1 g | $72.00 | |||
6-Chloro-1,3-dimethyluracil, 5 g | sc-214368A | 5 g | $106.00 |
