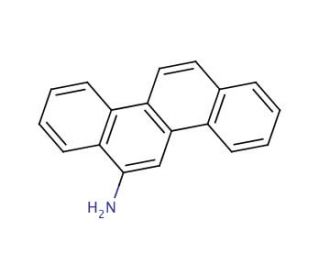
Molecular structure of 6-Aminochrysene, CAS Number: 2642-98-0
6-Aminochrysene (CAS 2642-98-0)
Alternate Names:
6-Chrysenamine
Application:
6-Aminochrysene is a potent inhibitor of transferase activity
CAS Number:
2642-98-0
Molecular Weight:
243.30
Molecular Formula:
C18H13N
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
6-Aminochrysene may functions as a potent carcinogen in experimental applications. It exerts its mechanism of action by binding to DNA, leading to the formation of DNA adducts. These adducts may cause mutations in the genetic material, ultimately contributing to the development of cancer. 6-Aminochrysene may induce oxidative stress and DNA damage, further promoting its carcinogenic effects. Its ability to disrupt normal cellular processes and promote genetic mutations may be a significant factor in the study of carcinogenesis.
6-Aminochrysene (CAS 2642-98-0) References
- Carcinogenicity of 6-aminochrysene in mice. | Lambelin, G., et al. 1975. Eur J Cancer (1965). 11: 327-34. PMID: 1171763
- Mutation induction and DNA adduct formation in Chinese hamster ovary cells treated with 6-nitrochrysene, 6-aminochrysene and their metabolites. | Delclos, KB. and Heflich, RH. 1992. Mutat Res. 279: 153-64. PMID: 1377330
- Identification of 6-aminochrysene photoproducts and study of the effect of a humic acid and riboflavin on its photolysis. | Zeng, K., et al. 2003. J Photochem Photobiol B. 72: 95-100. PMID: 14644571
- Activation of 6-aminochrysene to genotoxic products by different forms of rat liver cytochrome P450 in an O-acetyltransferase-overexpressing Salmonella typhimurium strain (NM2009). | Yamazaki, H. and Shimada, T. 1992. Biochem Pharmacol. 44: 913-20. PMID: 1530660
- Black tea intake modulates the excretion of urinary mutagens in rats exposed to 6-aminochrysene: induction of cytochromes P450 by 6-aminochrysene in the rat. | Yoxall, V., et al. 2005. Mutagenesis. 20: 23-8. PMID: 15598702
- Comparing cytotoxicity and genotoxicity in HaCaT cells caused by 6-aminochrysene and 5,6-chrysenequinone under ultraviolet A irradiation. | Zhang, Y., et al. 2006. Environ Toxicol Chem. 25: 1920-5. PMID: 16833155
- Preferential activation of 6-aminochrysene and 2-aminoanthracene to mutagenic moieties by different forms of cytochrome P450 in hepatic 9000 X g supernatants from the rat. | Lubet, RA., et al. 1989. Mutat Res. 212: 275-84. PMID: 2659983
- Metabolism of the carcinogen [3H]6-nitrochrysene in the preweanling mouse: identification of 6-aminochrysene-1,2-dihydrodiol as the probable proximate carcinogenic metabolite. | Delclos, KB., et al. 1988. Carcinogenesis. 9: 1875-84. PMID: 3168165
- Chrysene-Based Azahelicene π-Linker of D-π-D-Type Hole-Transporting Materials for Perovskite Solar Cells. | Tang, Z., et al. 2021. ChemSusChem. 14: 4923-4928. PMID: 34636480
- Identification of C8-modified deoxyinosine and N2- and C8-modified deoxyguanosine as major products of the in vitro reaction of N-hydroxy-6-aminochrysene with DNA and the formation of these adducts in isolated rat hepatocytes treated with 6-nitrochrysene and 6-aminochrysene. | Delclos, KB., et al. 1987. Carcinogenesis. 8: 1703-9. PMID: 3664962
- Stimulatory effect of 6-aminochrysene and congeners on zoxazolamine metabolism. | Donelli, MG. and Garattini, S. 1971. Biochem Pharmacol. 20: 2049-55. PMID: 5137960
- The antitumoral activity of some derivatives of 6-aminochrysene. | Tagnon, HJ., et al. 1970. Eur J Cancer (1965). 6: 81-7. PMID: 5450201
- Roles of different forms of cytochrome P450 in the activation of the promutagen 6-aminochrysene to genotoxic metabolites in human liver microsomes. | Yamazaki, H., et al. 1993. Carcinogenesis. 14: 1271-8. PMID: 8330339
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
6-Aminochrysene, 250 mg | sc-254871 | 250 mg | $131.00 |
