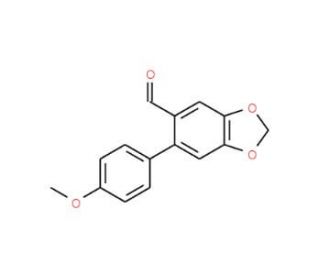
6-(4-Methoxyphenyl)-1,3-benzodioxole-5-carbaldehyde (CAS 875854-00-5)
QUICK LINKS
Methylmagnesium bromide (12% in Tetrahydrofuran, ca. 1mol/L) is a strong nucleophile used in organic synthesis. It functions as a source of the methyl anion, which can undergo nucleophilic addition reactions with a variety of electrophiles. Methylmagnesium bromide reacts with carbonyl compounds, such as ketones and aldehydes, to form alcohols through addition reactions. It can participate in Grignard reactions, where it adds to a variety of electrophiles, including alkyl halides, epoxides, and acid chlorides, to form new carbon-carbon bonds. Its mechanism of action involves the transfer of a methyl group to the electrophilic center of the substrate, leading to the formation of new carbon-carbon bonds and the generation of organic molecules with diverse structures.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
6-(4-Methoxyphenyl)-1,3-benzodioxole-5-carbaldehyde, 500 mg | sc-325666 | 500 mg | $198.00 | |||
6-(4-Methoxyphenyl)-1,3-benzodioxole-5-carbaldehyde, 1 g | sc-325666A | 1 g | $269.00 |
