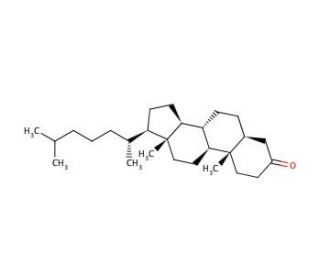
5α-Cholestan-3-one (CAS 566-88-1)
QUICK LINKS
5α-Cholestan-3-one, also referred to as 5α-dihydrotestosterone (DHT), plays a significant role in the intricate web of cholesterol and lipid metabolism. Derived from cholesterol, this compound participates in the processing of cholesterol, fatty acids, and various lipids within the body. 5α-Cholestan-3-one is recognized as a metabolite of testosterone. As a derivative of testosterone, The synthesis of 5α-Cholestan-3-one from cholesterol and its role as a key player in the metabolism of cholesterol, fatty acids, and lipids highlight its critical involvement in maintaining lipid homeostasis and regulating physiological functions. The presence and transformation of this compound within the body contribute to the intricate interplay of hormones and lipids.
5α-Cholestan-3-one (CAS 566-88-1) References
- Two moles of O2 consumption and one mole of H2O2 formation during cholesterol peroxidation with cholesterol oxidase from Pseudomonas sp. strain ST-200. | Doukyu, N. and Aono, R. 1999. Biochem J. 341 (Pt 3): 621-7. PMID: 10417325
- Synthesis and antimicrobial evaluation of some novel cholestane heterocyclic derivatives. | Elmegeed, GA., et al. 2004. Arch Pharm (Weinheim). 337: 140-7. PMID: 15038058
- Synthesis of thiazolyl and thieno cholestane derivatives: a novel class of potent antiinflammatory steroids. | Elmegeed, GA., et al. 2005. Pharmazie. 60: 328-33. PMID: 15918579
- Synthesis of [1,2,4]-triazolo-annulated 3-aza-A-homocholestanes--a novel class of pentacyclic compounds. | Bai, H., et al. 2012. Steroids. 77: 521-7. PMID: 22310161
- Effects of 5α-cholestan-3-one on the synaptic vesicle cycle at the mouse neuromuscular junction. | Kasimov, MR., et al. 2015. Biochim Biophys Acta. 1851: 674-85. PMID: 25725358
- Similar oxysterols may lead to opposite effects on synaptic transmission: Olesoxime versus 5α-cholestan-3-one at the frog neuromuscular junction. | Kasimov, MR., et al. 2016. Biochim Biophys Acta. 1861: 606-16. PMID: 27102612
- Oxysterol, 5α-cholestan-3-one, modulates a contractile response to β2-adrenoceptor stimulation in the mouse atria: Involvement of NO signaling. | Sytchev, VI., et al. 2017. Life Sci. 188: 131-140. PMID: 28888956
- Biosynthesis of cholestanol: 5-alpha-cholestan-3-one reductase of rat liver. | Shefer, S., et al. 1966. J Lipid Res. 7: 763-71. PMID: 4381999
- On the conversion of cholestanol into allocholic acid in rat liver. | Björkhem, I. and Gustafsson, J. 1971. Eur J Biochem. 18: 207-13. PMID: 5541504
- Investigation on the Favorable Formation of 5α-Cholestan-3α-ol and 5-Cholesten-3α-ol by Catalytic Hydrogenation | Ishige, M., & Shiota, M. 1975. Canadian Journal of Chemistry. 53(12): 1700-1707.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5α-Cholestan-3-one, 1 g | sc-214346 | 1 g | $121.00 | |||
5α-Cholestan-3-one, 5 g | sc-214346A | 5 g | $337.00 |
