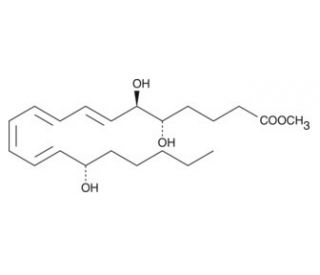
5(S),6(R)-Lipoxin A4 methyl ester (CAS 97643-35-1)
QUICK LINKS
5(S),6(R)-Lipoxin A4 methyl ester, with the CAS number 97643-35-1, is a chemically modified derivative of Lipoxin A4, a lipid mediator involved in the resolution of inflammation. This specific derivative features a methyl ester modification, which enhances its stability and alters its biological properties compared to its parent compound. The primary mechanism by which 5(S),6(R)-Lipoxin A4 methyl ester acts involves its interaction with specific cell surface receptors that are typically engaged by lipid mediators. This interaction can modulate various cellular pathways associated with inflammation, including the inhibition of leukocyte recruitment and the promotion of non-phlogistic phagocytosis by macrophages. In research, this compound has been utilized to study the extended effects and potential enhanced bioavailability provided by the methyl ester modification, particularly how these changes affect the molecule′s interaction with cellular receptors and subsequent signaling pathways. Studies focus on its role in synthetic pathways that mimic or influence natural anti-inflammatory processes, providing insights into the development of novel approaches for modulating inflammation without employing traditional methods. The insights gained from research involving 5(S),6(R)-Lipoxin A4 methyl ester are crucial for understanding the complex network of lipid mediators that govern the resolution phases of inflammatory responses.
5(S),6(R)-Lipoxin A4 methyl ester (CAS 97643-35-1) References
- Lipoxin A4 analogue protects brain and reduces inflammation in a rat model of focal cerebral ischemia reperfusion. | Ye, XH., et al. 2010. Brain Res. 1323: 174-83. PMID: 20138164
- Lipoxin A4 analog attenuates morphine antinociceptive tolerance, withdrawal-induced hyperalgesia, and glial reaction and cytokine expression in the spinal cord of rat. | Jin, H., et al. 2012. Neuroscience. 208: 1-10. PMID: 22366510
- Simvastatin and Benznidazole-Mediated Prevention of Trypanosoma cruzi-Induced Endothelial Activation: Role of 15-epi-lipoxin A4 in the Action of Simvastatin. | Campos-Estrada, C., et al. 2015. PLoS Negl Trop Dis. 9: e0003770. PMID: 25978361
- Emerging Role of Phospholipase-Derived Cleavage Products in Regulating Eosinophil Activity: Focus on Lysophospholipids, Polyunsaturated Fatty Acids and Eicosanoids. | Knuplez, E., et al. 2021. Int J Mol Sci. 22: PMID: 33919453
- Possibility of averting cytokine storm in SARS-COV 2 patients using specialized pro-resolving lipid mediators. | Yasmeen, N., et al. 2023. Biochem Pharmacol. 209: 115437. PMID: 36731803
- FoxO4 mediates macrophage M2 polarization by promoting LXA4R expression in an ovalbumin-induced allergic asthma model in mice. | Yu, T., et al. 2023. Allergol Immunopathol (Madr). 51: 19-30. PMID: 37422776
- The therapeutic potential of resolvins in pulmonary diseases. | Centanni, D., et al. 2023. Eur J Pharmacol. 958: 176047. PMID: 37742814
- Protective effects of Fufang Ejiao Jiang against aplastic anemia assessed by network pharmacology and metabolomics strategy | Dan, H., et al. 2021. Digital Chinese Medicine. 4(4): 328-342.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5(S),6(R)-Lipoxin A4 methyl ester, 25 µg | sc-205152 | 25 µg | $338.00 | |||
5(S),6(R)-Lipoxin A4 methyl ester, 50 µg | sc-205152A | 50 µg | $865.00 |
