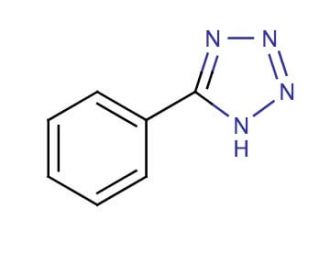
5-Phenyl-1H-tetrazole (CAS 18039-42-4)
QUICK LINKS
5-Phenyl-1H-tetrazole, a synthetic organic compound classified as a tetrazole, possesses an aromatic heterocyclic structure consisting of a five-membered ring comprising four nitrogen atoms and one carbon atom. Notably, it dissolves readily in water and ethanol, finding versatile applications in organic synthesis, particularly in the production of pharmaceuticals, agrochemicals, and various compounds. This compound, 5-Phenyl-1H-tetrazole, has been extensively employed in scientific research endeavors encompassing the synthesis of pharmaceuticals, agrochemicals, and diverse compounds. It has also played a role in generating novel heterocyclic compounds like imidazolium salts and polymers. Furthermore, its utility extends to the synthesis of metal complexes such as palladium and ruthenium, along with the production of biologically active compounds including antimicrobial agents and enzyme inhibitors. While the precise mechanism of action for 5-Phenyl-1H-tetrazole remains partially elucidated, it is believed to function as an electrophilic aromatic substitution reagent, reacting with nucleophiles like amines, alcohols, and carboxylic acids. Additionally, its properties as a Lewis acid enable reactions with electron-rich substrates such as enolates and enamines.
5-Phenyl-1H-tetrazole (CAS 18039-42-4) References
- Studies of the Antiproliferative Activity of Ruthenium (II) Cyclopentadienyl-Derived Complexes with Nitrogen Coordinated Ligands. | Moreno, V., et al. 2010. Bioinorg Chem Appl. 2010: PMID: 20689715
- Gas-phase behaviour of Ru(II) cyclopentadienyl-derived complexes with N-coordinated ligands by electrospray ionization mass spectrometry: fragmentation pathways and energetics. | Madeira, PJ., et al. 2012. Rapid Commun Mass Spectrom. 26: 1675-86. PMID: 22730088
- Self-assembly of a molecular crown as a structural analogue of calix[4]arene to modify Keggin anions. | Tian, A., et al. 2013. Dalton Trans. 42: 9809-12. PMID: 23698455
- Tetrazole-based, anhydrous proton exchange membranes for fuel cells. | Song, MK., et al. 2014. Adv Mater. 26: 1277-82. PMID: 24591010
- A new cube-based dodecanuclear cobalt(ii) cluster with azide and tetrazolate ligands exhibiting ferromagnetic ordering. | Zhang, XM., et al. 2015. Dalton Trans. 44: 13581-5. PMID: 26136165
- Enantioselective Denitrogenative Annulation of 1H-Tetrazoles with Styrenes Catalyzed by Rhodium. | Nakamuro, T., et al. 2018. Angew Chem Int Ed Engl. 57: 5497-5500. PMID: 29536660
- Magnetic chitosan stabilized Cu(II)-tetrazole complex: an effective nanocatalyst for the synthesis of 3-imino-2-phenylisoindolin-1-one derivatives under ultrasound irradiation. | Nasrollahzadeh, M., et al. 2022. Sci Rep. 12: 6724. PMID: 35468913
- Fe3O4@SiO2 nanoparticle supported ionic liquid for green synthesis of antibacterially active 1-carbamoyl-1-phenylureas in water. | Nasrollahzadeh, M., et al. 2018. RSC Adv. 8: 27631-27644. PMID: 35542743
- One-Pot Synthesis of 5-Substituted 1H-Tetrazoles from Aryl Bromides with Potassium Hexakis(cyano-κC)ferrate(4−) (K4[Fe(CN)6]) as Cyanide Source | and Yizhong Zhu, Yiming Ren, Chun Cai. 2009. Helvetica. 92: 171-175.
- Comparative Study on the Inhibition of Iron Corrosion in Aerated Stagnant 3.5 wt % Sodium Chloride Solutions by 5-Phenyl-1H-tetrazole and 3-Amino-1,2,4-triazole | Sherif, E. S. M. 2013. Industrial & Engineering Chemistry Research. 52: 14507-14513.
- A series of Keggin-based AgI-belt/cycle structures constructed from 5-phenyl-1H-tetrazole and its derivative through Ag–N and Ag–C bonds | Tian, A. X., Ji, X. B., Xiao, R., Ni, H. P., Tian, Y., Liu, G. C., & Ying, J. 2017. Journal of Coordination Chemistry,. 70: 404-416.
- Highly efficient three-component synthesis of 5-substituted-1H-tetrazoles from aldehydes, hydroxylamine, and tetrabutylammonium azide using doped nano-sized copper(I) oxide (Cu2O) on melamine–formaldehyde resin | Behrouz, S. 2017. Journal of Saudi Chemical Society. 21: 220-228.
- UV‐Induced 1, 3, 4‐Oxadiazole Formation from 5‐Substituted Tetrazoles and Carboxylic Acids in Flow | Green, L., Livingstone, K., Bertrand, S., Peace, S., & Jamieson, C. 2020. Chemistry–A European Journal. 26: 14866-14870.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Phenyl-1H-tetrazole, 25 g | sc-227007 | 25 g | $47.00 | |||
5-Phenyl-1H-tetrazole, 100 g | sc-227007A | 100 g | $100.00 | |||
5-Phenyl-1H-tetrazole, 1 kg | sc-227007B | 1 kg | $1000.00 |
