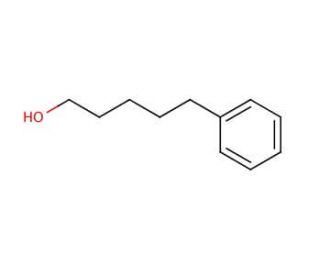
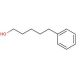
5-Phenyl-1-pentanol (CAS 10521-91-2)
QUICK LINKS
5-Phenyl-1-pentanol, a compound with a phenyl group attached to a pentanol backbone, has been extensively studied for its diverse range of applications in scientific research. One notable area of investigation is its role as a chiral building block in organic synthesis. The presence of the chiral center in the pentanol moiety renders 5-phenyl-1-pentanol a valuable starting material for the synthesis of enantiomerically pure compounds, particularly in asymmetric synthesis. Researchers exploit its chirality to catalyze stereoselective reactions, facilitating the production of pharmaceutical intermediates, natural products, and fine chemicals with high optical purity. Additionally, 5-phenyl-1-pentanol serves as a precursor in the synthesis of fragrances and flavoring agents due to its aromatic nature. Its ability to impart unique olfactory properties makes it a desirable candidate for the development of novel odorants and perfumes. Furthermore, 5-phenyl-1-pentanol has found utility as a ligand in coordination chemistry, participating in the formation of metal complexes with diverse structures and functionalities. Overall, the versatile nature of 5-phenyl-1-pentanol makes it an indispensable tool in organic synthesis, fragrance chemistry, and coordination chemistry research.
5-Phenyl-1-pentanol (CAS 10521-91-2) References
- Positional effect of solute functional group among positional isomers in hydroxyl group-solvent and carbonyl group-solvent specific interactions in menthanol--water mixed solvents monitored by high-performance liquid chromatography. | Cheong, WJ. and Keum, YI. 2001. J Chromatogr A. 910: 195-206. PMID: 11261714
- Aromatic alcohols and their effect on Gram-negative bacteria, cocci and mycobacteria. | Fraud, S., et al. 2003. J Antimicrob Chemother. 51: 1435-6. PMID: 12716779
- Synthesis of [131I/123I]-2-[5-(4-iodophenyl)pentyl]oxirane-2- carboxylic acid ethyl ester. | Abbas, HG., et al. 1991. Int J Rad Appl Instrum A. 42: 7-11. PMID: 1850395
- Repurposing approach identifies pitavastatin as a potent azole chemosensitizing agent effective against azole-resistant Candida species. | Eldesouky, HE., et al. 2020. Sci Rep. 10: 7525. PMID: 32372011
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Phenyl-1-pentanol, 1 g | sc-223702 | 1 g | $41.00 | |||
5-Phenyl-1-pentanol, 5 g | sc-223702A | 5 g | $131.00 |
