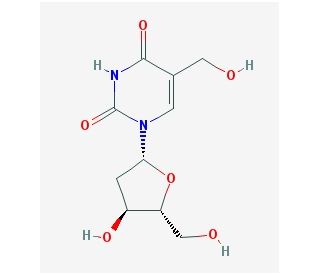
Molecular structure of 5-Hydroxymethyl-2′-deoxyuridine, CAS Number: 5116-24-5
5-Hydroxymethyl-2′-deoxyuridine (CAS 5116-24-5)
See product citations (1)
Alternate Names:
2′-Deoxy-5-hydroxymethyluridine; 2′-Deoxy-5-hydroxymethyl-D-uridine
Application:
5-Hydroxymethyl-2′-deoxyuridine is used as a marker for oxidative nucleic acid (DNA) damage
CAS Number:
5116-24-5
Purity:
≥98%
Molecular Weight:
258.23
Molecular Formula:
C10H14N2O6
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Hydroxymethyl-2′-deoxyuridine is a chemically modified nucleoside, closely related to the natural nucleosides found in DNA, but distinct due to a specific alteration in its structure. In genetic studies, 5-Hydroxymethyl-2′-deoxyuridine serves as an effective marker or probe. It helps scientists track genetic changes and study the incorporation and effects of different nucleosides in DNA, thereby contributing significantly to our understanding of genetic processes and mutations. Its modified structure, mirroring that of natural DNA but with a difference, underscores its utility in advancing our understanding of molecular biology.
5-Hydroxymethyl-2′-deoxyuridine (CAS 5116-24-5) References
- Effect of varying caloric restriction levels on female rat growth and 5-hydroxymethyl-2'-deoxyuridine in DNA. | Djuric, Z., et al. 2002. Toxicol Sci. 66: 125-30. PMID: 11861979
- Antibodies against 5-hydroxymethyl-2'-deoxyuridine are associated with lifestyle factors and GSTM1 genotype: a report from the Malmö Diet and Cancer cohort. | Wallström, P., et al. 2003. Cancer Epidemiol Biomarkers Prev. 12: 444-51. PMID: 12750240
- Measurement of the incorporation and repair of exogenous 5-hydroxymethyl-2'-deoxyuridine in human cells in culture using gas chromatography-negative chemical ionization-mass spectrometry. | Rogstad, DK., et al. 2007. Chem Res Toxicol. 20: 1787-96. PMID: 17914883
- Room temperature derivatization of 5-hydroxy-2'- deoxycytidine and 5-hydroxymethyl-2'-deoxyuridine for analysis by GC/MS. | Yu, F. and Djuric, Z. 1999. Biomarkers. 4: 85-92. PMID: 23898797
- 5-Hydroxymethyl-2'-deoxyuridine residues in the thrombin binding aptamer: investigating anticoagulant activity by making a tiny chemical modification. | Virgilio, A., et al. 2014. Chembiochem. 15: 2427-34. PMID: 25214456
- Evaluation of anti-HIV-1 mutagenic nucleoside analogues. | Vivet-Boudou, V., et al. 2015. J Biol Chem. 290: 371-83. PMID: 25398876
- Synergistic enhancement of 5-fluorouracil cytotoxicity by deoxyuridine analogs in cancer cells. | Matsumoto, Y., et al. 2015. Oncoscience. 2: 272-84. PMID: 25897430
- The oxidative damage to the human telomere: effects of 5-hydroxymethyl-2'-deoxyuridine on telomeric G-quadruplex structures. | Virgilio, A., et al. 2015. Org Biomol Chem. 13: 7421-9. PMID: 25997822
- Replacement of 2'-Deoxycytidine by 2'-Deoxycytidine Analogues in the E. coli Genome. | Mehta, AP., et al. 2016. J Am Chem Soc. 138: 14230-14233. PMID: 27762133
- The Multiple Cellular Roles of SMUG1 in Genome Maintenance and Cancer. | Raja, S. and Van Houten, B. 2021. Int J Mol Sci. 22: PMID: 33671338
- Diagnostic and Prognostic Power of Active DNA Demethylation Pathway Intermediates in Acute Myelogenous Leukemia and Myelodysplastic Syndromes. | Skalska-Bugala, A., et al. 2022. Cells. 11: PMID: 35269510
- UV-DDB stimulates the activity of SMUG1 during base excision repair of 5-hydroxymethyl-2'-deoxyuridine moieties. | Jang, S., et al. 2023. Nucleic Acids Res.. PMID: 36971122
- DNA base excision repair of 5-hydroxymethyl-2'-deoxyuridine stimulates poly(ADP-ribose) synthesis in Chinese hamster cells. | Boorstein, RJ., et al. 1995. Carcinogenesis. 16: 1173-9. PMID: 7767982
- Structures of base pairs with 5-(hydroxymethyl)-2'-deoxyuridine in DNA determined by NMR spectroscopy. | Mellac, S., et al. 1993. Biochemistry. 32: 7779-86. PMID: 8394115
- Molecular spectrum of mutations induced by 5-hydroxymethyl-2'-deoxyuridine in (CHO)-PL61 cells. | Chaung, W. and Boorstein, RJ. 1997. Mutat Res. 373: 125-37. PMID: 9015161
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Hydroxymethyl-2′-deoxyuridine, 25 mg | sc-221028 | 25 mg | $235.00 |
