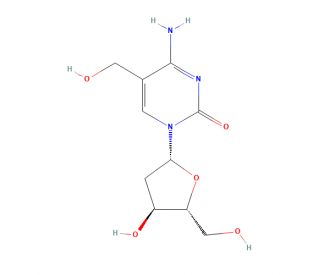
5-(Hydroxymethyl)-2′-deoxycytidine (CAS 7226-77-9)
See product citations (1)
QUICK LINKS
5-(Hydroxymethyl)-2′-deoxycytidine is a compound of interest in the field of molecular biology and genetics. It is a modified nucleoside that is incorporated into DNA during replication. Researchers study this compound to understand its influence on the stability and function of the DNA molecule, as well as its role in epigenetic modifications, which can affect gene expression without altering the underlying DNA sequence. In research applications, 5-(Hydroxymethyl)-2′-deoxycytidine is utilized to trace DNA methylation patterns and to investigate the mechanisms of DNA damage and repair. The hydroxymethyl group on the 5-carbon of the cytosine ring serves as a key marker in the study of DNA-protein interactions, where it can influence the binding of transcription factors and other DNA-associated proteins. Additionally, it is used in sequencing technologies to study the dynamics of the epigenome and to understand the cellular processes that are regulated by DNA methylation.
5-(Hydroxymethyl)-2′-deoxycytidine (CAS 7226-77-9) References
- Improved synthesis of 5-hydroxymethyl-2'-deoxycytidine phosphoramidite using a 2'-deoxyuridine to 2'-deoxycytidine conversion without temporary protecting groups. | Hansen, AS., et al. 2011. Bioorg Med Chem Lett. 21: 1181-4. PMID: 21256007
- Determination of genomic 5-hydroxymethyl-2'-deoxycytidine in human DNA by capillary electrophoresis with laser induced fluorescence. | Krais, AM., et al. 2011. Epigenetics. 6: 560-5. PMID: 21593596
- Deamination, oxidation, and C-C bond cleavage reactivity of 5-hydroxymethylcytosine, 5-formylcytosine, and 5-carboxycytosine. | Schiesser, S., et al. 2013. J Am Chem Soc. 135: 14593-9. PMID: 23980549
- DNA methylation dynamics in mouse preimplantation embryos revealed by mass spectrometry. | Okamoto, Y., et al. 2016. Sci Rep. 6: 19134. PMID: 26750605
- Associations of urinary 5-methyl-2'-deoxycytidine and 5-hydroxymethyl-2'-deoxycytidine with phthalate exposure and semen quality in 562 Chinese adult men. | Pan, Y., et al. 2016. Environ Int. 94: 583-590. PMID: 27346742
- Replacement of 2'-Deoxycytidine by 2'-Deoxycytidine Analogues in the E. coli Genome. | Mehta, AP., et al. 2016. J Am Chem Soc. 138: 14230-14233. PMID: 27762133
- Profiles of a broad spectrum of epigenetic DNA modifications in normal and malignant human cell lines: Proliferation rate is not the major factor responsible for the 5-hydroxymethyl-2'-deoxycytidine level in cultured cancerous cell lines. | Foksinski, M., et al. 2017. PLoS One. 12: e0188856. PMID: 29190698
- Enrichment and Quantitative Determination of 5-(Hydroxymethyl)-2'-deoxycytidine, 5-(Formyl)-2'-deoxycytidine, and 5-(Carboxyl)-2'-deoxycytidine in Human Urine of Breast Cancer Patients by Magnetic Hyper-Cross-Linked Microporous Polymers Based on Polyionic Liquid. | Guo, M., et al. 2018. Anal Chem. 90: 3906-3913. PMID: 29316399
- Characteristic profiles of DNA epigenetic modifications in colon cancer and its predisposing conditions-benign adenomas and inflammatory bowel disease. | Dziaman, T., et al. 2018. Clin Epigenetics. 10: 72. PMID: 29875879
- A novel malic acid-enhanced method for the analysis of 5-methyl-2'-deoxycytidine, 5-hydroxymethyl-2'-deoxycytidine, 5-methylcytidine and 5-hydroxymethylcytidine in human urine using hydrophilic interaction liquid chromatography-tandem mass spectrometry. | Guo, C., et al. 2018. Anal Chim Acta. 1034: 110-118. PMID: 30193624
- Active turnover of genomic methylcytosine in pluripotent cells. | Spada, F., et al. 2020. Nat Chem Biol. 16: 1411-1419. PMID: 32778844
- Photocaged 5-(Hydroxymethyl)pyrimidine Nucleoside Phosphoramidites for Specific Photoactivatable Epigenetic Labeling of DNA. | Chakrapani, A., et al. 2020. Org Lett. 22: 9081-9085. PMID: 33156631
- [Template Properties of 5-Methyl-2'-Deoxycytidine and 5-Hydroxymethyl-2'-Deoxycytidine in Reactions with Human Translesion and Reparative DNA Polymerases]. | Shilkin, ES., et al. 2021. Mol Biol (Mosk). 55: 305-311. PMID: 33871443
- XRCC1 Prevents Replication Fork Instability during Misincorporation of the DNA Demethylation Bases 5-Hydroxymethyl-2'-Deoxycytidine and 5-Hydroxymethyl-2'-Deoxyuridine. | Peña-Gómez, MJ., et al. 2022. Int J Mol Sci. 23: PMID: 35055077
- An Improved Approach for Practical Synthesis of 5-Hydroxymethyl-2'-deoxycytidine (5hmdC) Phosphoramidite and Triphosphate. | Yang, DZ., et al. 2022. Molecules. 27: PMID: 35164012
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-(Hydroxymethyl)-2′-deoxycytidine, 10 mg | sc-490666 | 10 mg | $393.00 |
