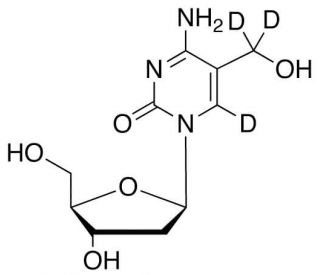
5-(Hydroxymethyl)-2′-deoxycytidine-d3 (CAS 7226-77-9 (unlabeled))
QUICK LINKS
5-(Hydroxymethyl)-2′-deoxycytidine-d3 is a deuterated analog of 5-(Hydroxymethyl)-2′-deoxycytidine, a compound that is used in the area of epigenetics and nucleic acids research. The incorporation of deuterium atoms increases the molecular stability and can be utilized to study the compound′s metabolism and dynamics within biological systems using mass spectrometry or NMR spectroscopy. This deuterated variant is particularly valuable in the examination of DNA modifications and repair mechanisms, as the hydroxymethyl group on the cytidine base is a known intermediate in the oxidation of 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine, both of which are involved in active DNA demethylation processes. By tracing the incorporation and fate of 5-(Hydroxymethyl)-2′-deoxycytidine-d3 in genomic DNA, researchers can gain insights into the role of hydroxymethylcytosine in gene expression regulation and the implications of DNA hydroxymethylation in development and disease.
5-(Hydroxymethyl)-2′-deoxycytidine-d3 (CAS 7226-77-9 (unlabeled)) References
- 5-Hydroxymethylcytosine is a predominantly stable DNA modification. | Bachman, M., et al. 2014. Nat Chem. 6: 1049-55. PMID: 25411882
- 5-Formylcytosine can be a stable DNA modification in mammals. | Bachman, M., et al. 2015. Nat Chem Biol. 11: 555-7. PMID: 26098680
- Single base resolution analysis of 5-hydroxymethylcytosine in 188 human genes: implications for hepatic gene expression. | Ivanov, M., et al. 2016. Nucleic Acids Res. 44: 6756-69. PMID: 27131363
- Differential DNA methylation at conserved non-genic elements and evidence for transgenerational inheritance following developmental exposure to mono(2-ethylhexyl) phthalate and 5-azacytidine in zebrafish. | Kamstra, JH., et al. 2017. Epigenetics Chromatin. 10: 20. PMID: 28413451
- Ascorbate regulates haematopoietic stem cell function and leukaemogenesis. | Agathocleous, M., et al. 2017. Nature. 549: 476-481. PMID: 28825709
- Higher Number of Night Shifts Associates with Good Perception of Work Capacity and Optimal Lung Function but Correlates with Increased Oxidative Damage and Telomere Attrition. | Pavanello, S., et al. 2019. Biomed Res Int. 2019: 8327629. PMID: 31111068
- DNA methylation changes involved in the tumor increase in F2 males born to gestationally arsenite-exposed F1 male mice. | Okamura, K., et al. 2019. Cancer Sci. 110: 2629-2642. PMID: 31215104
- Epigenetic, transcriptional and phenotypic responses in two generations of Daphnia magna exposed to the DNA methylation inhibitor 5-azacytidine. | Lindeman, LC., et al. 2019. Environ Epigenet. 5: dvz016. PMID: 31528364
- Global DNA methylation levels in white blood cells of patients with chronic heroin use disorder. A prospective study. | Fragou, D., et al. 2021. Toxicol Rep. 8: 337-342. PMID: 33643851
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-(Hydroxymethyl)-2′-deoxycytidine-d3, 1 mg | sc-495314 | 1 mg | $516.00 |
