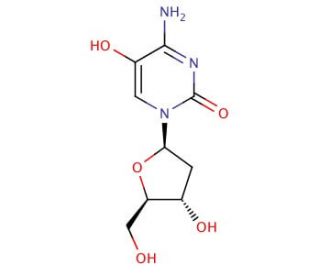
Molecular structure of 5-Hydroxy-2′-deoxycytidine
5-Hydroxy-2′-deoxycytidine (CAS 52278-77-0)
Alternate Names:
2-Deoxy-5-hydroxy-cytidine
Application:
5-Hydroxy-2′-deoxycytidine is 5-Hydroxy-2′-deoxycytidine is an abnormal base residue in cellular DNA.
CAS Number:
52278-77-0
Molecular Weight:
243.22
Molecular Formula:
C9H13N3O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Hydroxy-2′-deoxycytidine is a modified nucleoside that consists of the nucleobase cytosine linked to the sugar deoxyribose, with a hydroxyl group (-OH) attached to the 5th carbon of the sugar. It is derived from the normal DNA nucleoside 2′-deoxycytidine through oxidation at the 5th carbon. This modified nucleoside is involved in various biological processes and has been studied for its potential role in DNA repair and epigenetic regulation. It is also considered a biomarker for oxidative stress and DNA damage.
5-Hydroxy-2′-deoxycytidine (CAS 52278-77-0) References
- Identification by UV resonance Raman spectroscopy of an imino tautomer of 5-hydroxy-2'-deoxycytidine, a powerful base analog transition mutagen with a much higher unfavored tautomer frequency than that of the natural residue 2'-deoxycytidine. | Suen, W., et al. 1999. Proc Natl Acad Sci U S A. 96: 4500-5. PMID: 10200291
- Glutathione and ascorbate are negatively correlated with oxidative DNA damage in human lymphocytes. | Lenton, KJ., et al. 1999. Carcinogenesis. 20: 607-13. PMID: 10223188
- Oxidation of 5-hydroxypyrimidine nucleosides to 5-hydroxyhydantoin and its alpha-hydroxy-ketone isomer. | Rivière, J., et al. 2005. Chem Res Toxicol. 18: 1332-8. PMID: 16097807
- Collisionally activated dissociation of protonated 2'-deoxycytidine, 2'-deoxyuridine, and their oxidatively damaged derivatives. | Cao, H. and Wang, Y. 2006. J Am Soc Mass Spectrom. 17: 1335-1341. PMID: 16872831
- Major oxidative products of cytosine are substrates for the nucleotide incision repair pathway. | Daviet, S., et al. 2007. DNA Repair (Amst). 6: 8-18. PMID: 16978929
- Oxidation of 2'-deoxycytidine to four interconverting diastereomers of N1-carbamoyl-4,5-dihydroxy-2-oxoimidazolidine nucleosides. | Tremblay, S., et al. 2007. J Org Chem. 72: 3672-8. PMID: 17439282
- Reaction of 2'-deoxycytidine with peroxynitrite in the presence of ammonium bromide. | Suzuki, T., et al. 2008. Bioorg Med Chem. 16: 5164-70. PMID: 18358730
- Defective repair of 5-hydroxy-2'-deoxycytidine in Cockayne syndrome cells and its complementation by Escherichia coli formamidopyrimidine DNA glycosylase and endonuclease III. | Foresta, M., et al. 2010. Free Radic Biol Med. 48: 681-90. PMID: 20026203
- Mutagen-mediated enhancement of HIV-1 replication in persistently infected cells. | Sánchez-Jiménez, C., et al. 2012. Virology. 424: 147-53. PMID: 22265575
- Thermodynamic signature of DNA damage: characterization of DNA with a 5-hydroxy-2'-deoxycytidine·2'-deoxyguanosine base pair. | Ganguly, M., et al. 2012. Biochemistry. 51: 2018-27. PMID: 22332945
- Effects of halides on reaction of nucleosides with ozone. | Suzuki, T., et al. 2012. Nucleosides Nucleotides Nucleic Acids. 31: 461-73. PMID: 22646086
- Human NEIL3 is mainly a monofunctional DNA glycosylase removing spiroimindiohydantoin and guanidinohydantoin. | Krokeide, SZ., et al. 2013. DNA Repair (Amst). 12: 1159-64. PMID: 23755964
- Room temperature derivatization of 5-hydroxy-2'- deoxycytidine and 5-hydroxymethyl-2'-deoxyuridine for analysis by GC/MS. | Yu, F. and Djuric, Z. 1999. Biomarkers. 4: 85-92. PMID: 23898797
- New substrates for old enzymes. 5-Hydroxy-2'-deoxycytidine and 5-hydroxy-2'-deoxyuridine are substrates for Escherichia coli endonuclease III and formamidopyrimidine DNA N-glycosylase, while 5-hydroxy-2'-deoxyuridine is a substrate for uracil DNA N-glycosylase. | Hatahet, Z., et al. 1994. J Biol Chem. 269: 18814-20. PMID: 8034633
- Synthesis of oligonucleotides containing two putatively mutagenic DNA lesions: 5-hydroxy-2'-deoxyuridine and 5-hydroxy-2'-deoxycytidine. | Morningstar, ML., et al. 1997. Chem Res Toxicol. 10: 1345-50. PMID: 9437524
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Hydroxy-2′-deoxycytidine, 2 mg | sc-490884A | 2 mg | $89.00 | |||
5-Hydroxy-2′-deoxycytidine, 10 mg | sc-490884 | 10 mg | $317.00 | |||
5-Hydroxy-2′-deoxycytidine, 50 mg | sc-490884B | 50 mg | $1202.00 |
