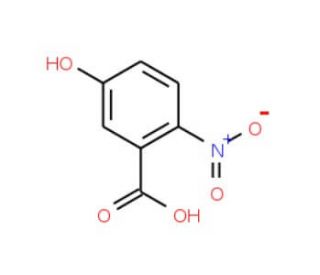
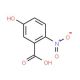
5-Hydroxy-2-nitrobenzoic acid (CAS 610-37-7)
QUICK LINKS
5-Hydroxy-2-nitrobenzoic acid is a distinct aromatic compound characterized by the presence of a nitro group at the second position and a hydroxy group at the fifth position on the benzene ring, anchored by a carboxylic acid group. The nitro group, being a strong electron-withdrawing group, significantly influences the electronic structure of the benzene ring, enhancing the electrophilic nature of the carboxyl group, while the electron-donating hydroxy group introduces additional resonance stability. This unique arrangement of substituents makes the compound an important subject in chemical research, particularly for studying the effects of substituent positioning on reactivity and stability within aromatic systems. It is extensively used as a synthetic intermediate in the production of complex organic molecules, including dyes and pharmaceutical intermediates. In synthetic chemistry, 5-Hydroxy-2-nitrobenzoic acid facilitates diverse reactions such as esterification and nitration, providing insights into reaction dynamics and mechanisms. Additionally, the compound serves as a model for exploring fundamental concepts in organic chemistry such as electron donation and withdrawal, resonance effects, and the impact of functional groups on acid-base behavior, making it a valuable tool in both academic and applied research settings.
5-Hydroxy-2-nitrobenzoic acid (CAS 610-37-7) References
- The decomposition of 2,5-dinitrobenzoic acid by alkali. | LANGLEY, WD. 1948. J Am Chem Soc. 70: 1633. PMID: 18915783
- Omega-conotoxin GVIA mimetics based on an anthranilamide core: effect of variation in ammonium side chain lengths and incorporation of fluorine. | Andersson, A., et al. 2009. Bioorg Med Chem. 17: 6659-70. PMID: 19683451
- Synthesis, properties, and remarkable 2 D Self-Assembly at the Liquid/Solid interface of a series of triskele-shaped 5,11,17-triazatrinaphthylenes (TrisK). | Bertrand, H., et al. 2013. Chemistry. 19: 14654-64. PMID: 24026879
- 3-Hydroxy-2-Nitrobenzoic Acid as a MALDI Matrix for In-Source Decay and Evaluation of the Isomers. | Fukuyama, Y., et al. 2018. J Am Soc Mass Spectrom. 29: 2227-2236. PMID: 30062476
- On the Search of a Silver Bullet for the Preparation of Bioinspired Molecular Electrets with Propensity to Transfer Holes at High Potentials. | Derr, JB., et al. 2021. Biomolecules. 11: PMID: 33804209
- Comparative metabolism of nitroaromatic compounds in freshwater, brackish water and marine decapod crustaceans. | Foster, GD. and Crosby, DG. 1987. Xenobiotica. 17: 1393-404. PMID: 3439190
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Hydroxy-2-nitrobenzoic acid, 1 g | sc-268076 | 1 g | $92.00 |
