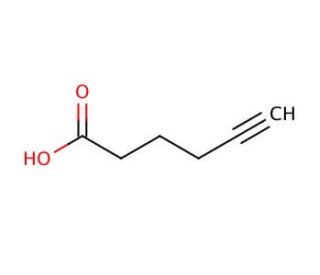
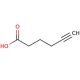
5-Hexynoic acid (CAS 53293-00-8)
QUICK LINKS
5-Hexynoic acid is an inhibitor of certain enzymatic processes. It acts by binding to specific enzyme active sites, disrupting the normal catalytic function and leading to the inhibition of the enzyme′s activity. The compound′s mechanism of action involves the formation of covalent bonds with the target enzyme, altering its structure and preventing the substrate from binding effectively. This interference with enzymatic activity can be utilized to study the specific pathways and processes in which the enzyme is involved, providing insights into the underlying biochemical mechanisms. In experimental applications, 5-Hexynoic acid′s inhibitory properties can be harnessed to investigate the role of the targeted enzyme in various cellular and molecular processes, contributing to a deeper understanding of the biological systems under study.
5-Hexynoic acid (CAS 53293-00-8) References
- Heterofunctional polymers and core-shell nanoparticles via cascade aminolysis/Michael addition and alkyne-azide click reaction of RAFT polymers. | An, Z., et al. 2008. Chem Commun (Camb). 6501-3. PMID: 19057759
- Versatility of alkyne-modified poly(glycidyl methacrylate) layers for click reactions. | Soto-Cantu, E., et al. 2011. Langmuir. 27: 5986-96. PMID: 21506527
- Development of endocannabinoid-based chemical probes for the study of cannabinoid receptors. | Martín-Couce, L., et al. 2011. J Med Chem. 54: 5265-9. PMID: 21675776
- Cyclization of 5-hexynoic acid to 3-alkoxy-2-cyclohexenones. | Hylden, AT., et al. 2011. Beilstein J Org Chem. 7: 1323-6. PMID: 22043242
- A versatile method for functionalization and grafting of 2-hydroxyethyl cellulose (HEC) via Click chemistry. | Eissa, AM., et al. 2012. Carbohydr Polym. 90: 859-69. PMID: 22840013
- Single 'click' synthesis of a mixed-mode silica sorbent and application in matrix solid-phase dispersion extraction of β-agonists from porcine liver. | Zhu, Y., et al. 2014. J Chromatogr A. 1354: 101-8. PMID: 24929910
- Mixed alkyl aryl phosphonate esters as quenched fluorescent activity-based probes for serine proteases. | Serim, S., et al. 2015. Org Biomol Chem. 13: 2293-9. PMID: 25553959
- Configurable Nanosized Metal Oxide Oligomers via Precise 'Click' Coupling Control of Hybrid Polyoxometalates. | Macdonell, A., et al. 2015. J Am Chem Soc. 137: 5662-5. PMID: 25846763
- Controlled Chemical Derivatisation of Carbon Nanotubes with Imaging, Targeting, and Therapeutic Capabilities. | Ménard-Moyon, C., et al. 2015. Chemistry. 21: 14886-92. PMID: 26331300
- Synthesis and biological evaluation of PSMA-targeting paclitaxel conjugates. | Machulkin, AE., et al. 2019. Bioorg Med Chem Lett. 29: 2229-2235. PMID: 31248772
- Redox-Switchable Cycloisomerization of Alkynoic Acids with Napthalenediimide-Derived N-Heterocyclic Carbene Complexes. | Ruiz-Zambrana, C., et al. 2021. Angew Chem Int Ed Engl. 60: 20003-20011. PMID: 34255909
- A bioorthogonal chemical reporter for fatty acid synthase-dependent protein acylation. | Karthigeyan, KP., et al. 2021. J Biol Chem. 297: 101272. PMID: 34606827
- Single-component nanodiscs via the thermal folding of amphiphilic graft copolymers with the adjusted flexibility of the main chain. | Nishimura, T., et al. 2022. Chem Sci. 13: 5243-5251. PMID: 35655565
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Hexynoic acid, 5 g | sc-239046 | 5 g | $120.00 |
