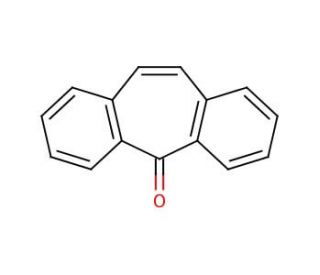
Molecular structure of 5-Dibenzosuberenone, CAS Number: 2222-33-5
5-Dibenzosuberenone (CAS 2222-33-5)
Alternate Names:
5H-Dibenzo[a,d]cyclohepten-5-one
CAS Number:
2222-33-5
Purity:
≥98%
Molecular Weight:
206.24
Molecular Formula:
C15H10O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Dibenzosuberenone functions as a reactive intermediate in organic synthesis. It acts as a key building block in the formation of various organic compounds, serving as a precursor in the synthesis of complex molecules. Its mechanism of action involves participating in a range of chemical reactions, including cyclization, oxidation, and functional group transformations. 5-Dibenzosuberenone exhibits reactivity towards a variety of nucleophiles and electrophiles, allowing for the formation of diverse chemical structures. Its role in the experiment involves facilitating the creation of new chemical entities with potential applications in materials science and agrochemicals.
5-Dibenzosuberenone (CAS 2222-33-5) References
- Zinc(II)-catalyzed addition of Grignard reagents to ketones. | Hatano, M., et al. 2010. J Org Chem. 75: 5008-16. PMID: 20560525
- Synthesis of 1,2-Dioxetanes as Thermochemiluminescent Labels for Ultrasensitive Bioassays: Rational Prediction of Olefin Photooxygenation Outcome by Using a Chemometric Approach. | Andronico, LA., et al. 2016. Chemistry. 22: 18156-18168. PMID: 27798823
- Pd(II)-Catalyzed Direct ortho-C-H Acylation of Aromatic Ketones by Oxidative Decarboxylation of α-Oxocarboxylic Acids. | Lee, PY., et al. 2017. Org Lett. 19: 2082-2085. PMID: 28374587
- From UTP to AR-C118925, the discovery of a potent non nucleotide antagonist of the P2Y2 receptor. | Kindon, N., et al. 2017. Bioorg Med Chem Lett. 27: 4849-4853. PMID: 28958619
- Fluorinated bisbenzimidazoles: a new class of drug-like anion transporters with chloride-mediated, cell apoptosis-inducing activity. | Yu, XH., et al. 2019. Org Biomol Chem. 17: 1558-1571. PMID: 30694281
- Combustion in the future: The importance of chemistry. | Kohse-Höinghaus, K. 2020. Proc Combust Inst.. PMID: 33013234
- Nitrosoarene-Catalyzed HFIP-Assisted Transformation of Arylmethyl Halides to Aromatic Carbonyls under Aerobic Conditions. | Pradhan, S., et al. 2021. Org Lett. 23: 6148-6152. PMID: 34284588
- Catalytic cyclopropanation reactions with α-silyl-, germanyl- and stannyl carbenes generated from cyclopropenes. | Coto, D., et al. 2022. Chem Commun (Camb). 58: 8416-8419. PMID: 35796243
- Enantioselective Copper-Catalyzed Formal [2+1] and [4+1] Annulations of Diynes with Ketones via Carbonyl Ylides. | Qi, LJ., et al. 2022. Angew Chem Int Ed Engl. 61: e202210637. PMID: 35975959
- Direct nucleophilic and electrophilic activation of alcohols using a unified boron-based organocatalyst scaffold. | Rygus, JPG. and Hall, DG. 2023. Nat Commun. 14: 2563. PMID: 37142592
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Dibenzosuberenone, 25 g | sc-233347 | 25 g | $68.00 |
