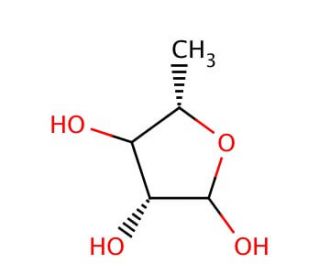
5-Deoxy-L-arabinose (CAS 13039-56-0)
QUICK LINKS
5-Deoxy-L-arabinose is a rare sugar with distinctive structural features that make it a valuable compound for scientific research, particularly in the fields of carbohydrate chemistry and microbial metabolism. This sugar lacks the hydroxyl group at the 5-position compared to its parent compound L-arabinose, altering its chemical reactivity and biological interactions. The modification in its structure confers unique properties that are exploited in various research applications. 5-Deoxy-L-arabinose has been used extensively to study bacterial pathways that involve the catabolism of rare sugars, as some bacteria possess specific enzymes capable of metabolizing such sugars, revealing potential pathways for biotechnological applications, such as biofuel production and bioremediation. Additionally, this sugar is used in synthetic chemistry to explore new methods of glycosylation and oligosaccharide assembly, where its lack of a 5-hydroxyl group can lead to novel branching patterns or termination points in polysaccharide chains. This research helps in understanding how changes in sugar structure affect their biological function and stability, contributing to advances in the synthesis of complex carbohydrate-based materials. Thus, 5-Deoxy-L-arabinose serves as a crucial tool in applications, offering insights into carbohydrate biochemistry and microbial ecology.
5-Deoxy-L-arabinose (CAS 13039-56-0) References
- Pteridines. XXXVII. A total synthesis of L-erythro-biopterin and some related 6-(polyhydroxyalkyl)pterins. | Taylor, EC. and Jacobi, PA. 1976. J Am Chem Soc. 98: 2301-7. PMID: 1254867
- Indirect approach to C-3 branched 1,2-cis-glycofuranosides: synthesis of aceric acid glycoside analogues. | de Oliveira, MT., et al. 2008. Carbohydr Res. 343: 211-20. PMID: 18039541
- Highly α-selective hydrolysis of α,β-epoxyalcohols using tetrabutylammonium fluoride. | Mukerjee, P., et al. 2010. Org Lett. 12: 3986-9. PMID: 20722386
- Pectin-modifying enzymes and pectin-derived materials: applications and impacts. | Bonnin, E., et al. 2014. Appl Microbiol Biotechnol. 98: 519-32. PMID: 24270894
- Secondary metabolites from the fermented rice of the fungus Monascus purpureus and their bioactivities. | Wu, HC., et al. 2019. Nat Prod Res. 33: 3541-3550. PMID: 30518252
- Strong inhibitory effect of furanoses and sugar lactones on beta-galactosidase Escherichia coli. | Huber, RE. and Brockbank, RL. 1987. Biochemistry. 26: 1526-31. PMID: 3109465
- Hybrid Chemoenzymatic Synthesis of C7 -Sugars for Molecular Evidence of in vivo Shikimate Pathway Inhibition. | Rath, P., et al. 2022. Chembiochem. 23: e202200241. PMID: 35508894
- Letter: Transition metal ion inhibition of enzyme-catalyzed phosphate ester displacement reactions. | VanEtten, RL., et al. 1974. J Am Chem Soc. 96: 6782-5. PMID: 4414446
- 13C and 15N nuclear magnetic resonance evidence that the active site carboxyl group of dihydrofolate reductase is not involved in the relay of a proton to substrate. | Blakley, RL., et al. 1993. Arch Biochem Biophys. 306: 501-9. PMID: 8105754
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Deoxy-L-arabinose, 250 mg | sc-221023 | 250 mg | $360.00 |
