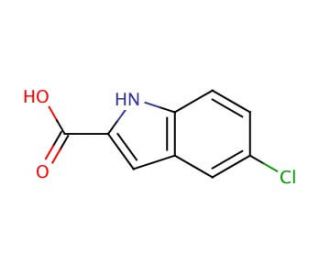
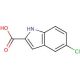
5-Chloroindole-2-carboxylic acid (CAS 10517-21-2)
QUICK LINKS
5-Chloroindole-2-carboxylic acid is a compound that functions as a key intermediate in the synthesis of various indole derivatives. It plays a role in the development of novel compounds with potential biological activity. Involved in the modification of indole-based molecules, allowing for the creation of diverse chemical structures for purposes. Its mode of action involves participating in pathways to produce indole-based compounds with specific structural features. By serving as a building block in the synthesis of new molecules, it contributes to the exploration of structure-activity relationships and the investigation of potential biological effects. At the molecular level, 5-Chloroindole-2-carboxylic acid undergoes chemical reactions to introduce specific functional groups, enabling the creation of diverse chemical entities for experimental investigation. Its involvement in processes allows for the generation of a range of indole derivatives for development purposes.
5-Chloroindole-2-carboxylic acid (CAS 10517-21-2) References
- Specificity and potency of N-methyl-D-aspartate glycine site antagonists and of mephenesin on the rat spinal cord in vitro. | Pralong, E., et al. 1992. Neurosci Lett. 136: 56-8. PMID: 1378953
- Novel 3,4-dihydroquinolin-2(1H)-one inhibitors of human glycogen phosphorylase a. | Rosauer, KG., et al. 2003. Bioorg Med Chem Lett. 13: 4385-8. PMID: 14643331
- Cycloalkanediamine derivatives as novel blood coagulation factor Xa inhibitors. | Nagata, T., et al. 2007. Bioorg Med Chem Lett. 17: 4683-8. PMID: 17555959
- Antihyperlipidemic properties of novel N-(benzoylphenyl)-5-substituted-1H-indole-2-carboxamides in Triton WR-1339-induced hyperlipidemic rats. | Al-Hiari, Y., et al. 2011. Molecules. 16: 8292-304. PMID: 21959300
- Structure-activity relationship study of indole-2-carboxamides identifies a potent allosteric modulator for the cannabinoid receptor 1 (CB1). | Mahmoud, MM., et al. 2013. J Med Chem. 56: 7965-75. PMID: 24053617
- Synthesis and biological evaluation of indole-2-carboxamides bearing photoactivatable functionalities as novel allosteric modulators for the cannabinoid CB1 receptor. | Qiao, CJ., et al. 2016. Eur J Med Chem. 121: 517-529. PMID: 27318976
- Immunological studies of sporidesmin: production of antibodies to azo-linked derivatives of 2-amino-5-chloro-3,4-dimethoxy benzyl alcohol. | Jonas, WE. and Erasmuson, AF. 1977. N Z Vet J. 25: 161-4. PMID: 275713
- Design, Synthesis, and Use of Novel Photoaffinity Probes in Measuring the Serum Concentration of Glycogen Phosphorylase. | Zhang, Y., et al. 2019. Molecules. 24: PMID: 30813328
- Optimization of Peptidomimetics as Selective Inhibitors for the β-Catenin/T-Cell Factor Protein-Protein Interaction. | Wang, Z., et al. 2019. J Med Chem. 62: 3617-3635. PMID: 30856332
- Design, synthesis, biological evaluation and docking study of novel indole-2-amide as anti-inflammatory agents with dual inhibition of COX and 5-LOX. | Huang, Y., et al. 2019. Eur J Med Chem. 180: 41-50. PMID: 31299586
- Synthesis, in vitro ADME profiling and in vivo pharmacological evaluation of novel glycogen phosphorylase inhibitors. | Miao, GX., et al. 2020. Bioorg Med Chem Lett. 30: 127117. PMID: 32527535
- Design, synthesis and antimycobacterial evaluation of novel adamantane and adamantanol analogues effective against drug-resistant tuberculosis. | Alsayed, SSR., et al. 2021. Bioorg Chem. 106: 104486. PMID: 33276981
- Hydrogen-Bonding Ability of Noyori-Ikariya Catalysts Enables Stereoselective Access to CF3-Substituted syn-1,2-Diols via Dynamic Kinetic Resolution. | Sterle, M., et al. 2023. ACS Catal. 13: 6242-6248. PMID: 37180962
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Chloroindole-2-carboxylic acid, 1 g | sc-254817 | 1 g | $31.00 |
