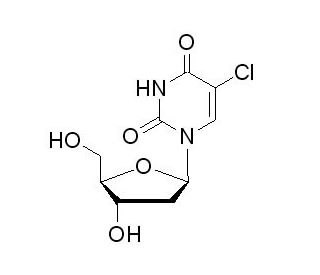
Molecular structure of 5-Chloro-2′-deoxyuridine, CAS Number: 50-90-8
5-Chloro-2′-deoxyuridine (CAS 50-90-8)
Alternate Names:
CldU
Application:
5-Chloro-2′-deoxyuridine is a thymidine analogue
CAS Number:
50-90-8
Molecular Weight:
262.65
Molecular Formula:
C9H11ClN2O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Chloro-2′-deoxyuridine (5-Cl-dUrd, CldU) is a halogenated uridine derivative employed in studying DNA and DNA precursor damage. It serves as a thymidine analogue and is readily incorporated into newly synthesized DNA in place of thymidine following phosphorylation. The presence of thymidine analogs, including CldU, can induce significant modifications in DNA processing and replication, making them useful as mutagens, clastogens, and antiviral agents in various applications.
5-Chloro-2′-deoxyuridine (CAS 50-90-8) References
- Vibrational study of a nucleoside analogue with antiviral activity, 5-chloro-2'-deoxyuridine, CDU. | Bailey, L., et al. 1999. Nucleosides Nucleotides. 18: 1069-71. PMID: 10432741
- 5-chloro-2'-deoxyuridine cytotoxicity results from base excision repair of uracil subsequent to thymidylate synthase inhibition. | Brandon, ML., et al. 2000. Mutat Res. 459: 161-9. PMID: 10725666
- Analytical and pharmacokinetic studies with 5-chloro-2'-deoxycytidine. | Hale, JT., et al. 2002. Biochem Pharmacol. 64: 1493-502. PMID: 12417262
- Inhibition of poly(ADP-ribose)polymerase activity by nucleoside analogs of thymidine. | Pivazyan, AD., et al. 1992. Biochem Pharmacol. 44: 947-53. PMID: 1530662
- Metal ion-binding properties of (N3)-deprotonated uridine, thymidine, and related pyrimidine nucleosides in aqueous solution. | Knobloch, B., et al. 2005. Proc Natl Acad Sci U S A. 102: 7459-64. PMID: 15897459
- Discrimination of cell nuclei in early S-phase, mid-to-late S-phase, and G(2)/M-phase by sequential administration of 5-bromo-2'-deoxyuridine and 5-chloro-2'-deoxyuridine. | Yamada, K., et al. 2005. J Histochem Cytochem. 53: 1365-70. PMID: 15956030
- Chemical Morphing of DNA Containing Four Noncanonical Bases. | Eremeeva, E., et al. 2016. Angew Chem Int Ed Engl. 55: 7515-9. PMID: 27159019
- DNA Fiber Spreading Assay to Test HDACi Effects on DNA and Its Replication. | Nikolova, T., et al. 2017. Methods Mol Biol. 1510: 103-113. PMID: 27761816
- The 5-chlorouracil:7-deazaadenine base pair as an alternative to the dT:dA base pair. | Eremeeva, E., et al. 2016. Org Biomol Chem. 15: 168-176. PMID: 27918055
- Modulation of BACE1 Activity by Chemically Modified Aptamers. | Gasse, C., et al. 2018. Chembiochem. 19: 754-763. PMID: 29327496
- The Olfactory Bulb Provides a Radioresistant Niche for Glioblastoma Cells. | Timme, CR., et al. 2020. Int J Radiat Oncol Biol Phys. 107: 194-201. PMID: 31987963
- Single Molecular Resolution to Monitor DNA Replication Fork Dynamics upon Stress by DNA Fiber Assay. | Liu, W. 2021. Bio Protoc. 11: e4269. PMID: 35087928
- A novel thymidine phosphorylase to synthesize (halogenated) anticancer and antiviral nucleoside drugs in continuous flow. | Benítez-Mateos, AI., et al. 2022. Catal Sci Technol. 12: 6231-6238. PMID: 36325519
- Plasticity of cell proliferation in the retina of Austrolebias charrua fish under light and darkness conditions. | Berrosteguieta, I., et al. 2022. Curr Res Neurobiol. 3: 100042. PMID: 36518338
- The induction of specific-locus mutations and sister-chromatid exchanges by 5-bromo- and 5-chloro-deoxyuridine. | Heartlein, MW., et al. 1982. Mutat Res. 92: 411-6. PMID: 7201072
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Chloro-2′-deoxyuridine, 100 mg | sc-221018 | 100 mg | $104.00 |
