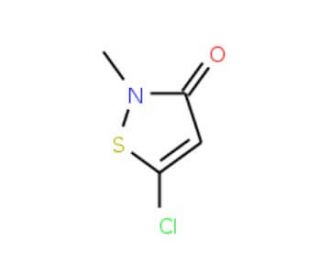
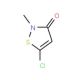
Molecular structure of 5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0), CAS Number: 26172-55-4
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) (CAS 26172-55-4)
See product citations (5)
Alternate Names:
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) is also known as Methylchloroisothiazolinone.
Application:
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) is an isothiazolinone and preservative agent with antimicrobial and antifungal properties.
CAS Number:
26172-55-4
Purity:
Active Ingredient >14%
Molecular Weight:
149.60
Molecular Formula:
C4H4ClNOS
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) is an isothiazolinone. The active sulphur moiety of 5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) is able to oxidize thiol-containing residues, effectively killing most aerobic and anaerobic bacteria. In addition to its antimicrobial properties, it has antifungal properties as well, and is found in many water-based personal care products. 5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) may be used in conjunction with other preservatives such as ethylparaben or benzalkonium chloride. It may also be used in the production of glue, detergents, paints, and fuels.
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0) (CAS 26172-55-4) References
- Preservation of products with MCI/MI in Switzerland. | Reinhard, E., et al. 2001. Contact Dermatitis. 45: 257-64. PMID: 11722483
- In vitro induction of apoptosis vs. necrosis by widely used preservatives: 2-phenoxyethanol, a mixture of isothiazolinones, imidazolidinyl urea and 1,2-pentanediol. | Anselmi, C., et al. 2002. Biochem Pharmacol. 63: 437-53. PMID: 11853695
- Involvement of oxidative stress in apoptosis induced by a mixture of isothiazolinones in normal human keratinocytes. | Ettorre, A., et al. 2003. J Invest Dermatol. 121: 328-36. PMID: 12880425
- GSH depletion, protein S-glutathionylation and mitochondrial transmembrane potential hyperpolarization are early events in initiation of cell death induced by a mixture of isothiazolinones in HL60 cells. | Di Stefano, A., et al. 2006. Biochim Biophys Acta. 1763: 214-25. PMID: 16458373
- Iron dependent degradation of an isothiazolone biocide (5-chloro-2-methyl-4-isothiazolin-3-one). | Tanji, Y., et al. 2007. Biofouling. 23: 73-7. PMID: 17453731
- Role of intracellular calcium and S-glutathionylation in cell death induced by a mixture of isothiazolinones in HL60 cells. | Frosali, S., et al. 2009. Biochim Biophys Acta. 1793: 572-83. PMID: 19118583
- Growth inhibitory and biocidal activity of some isothiazolone biocides. | Collier, PJ., et al. 1990. J Appl Bacteriol. 69: 569-77. PMID: 2292520
- [Kathon CG--a new contact sensitizing preservative]. | Knudsen, BB. and Menné, T. 1990. Ugeskr Laeger. 152: 656-7. PMID: 2321281
- Analysis of isothiazolinone preservatives in polyvinyl alcohol cooling towels used in Japan. | Kawakami, T., et al. 2014. J Environ Sci Health A Tox Hazard Subst Environ Eng. 49: 1209-17. PMID: 24967553
- Photoaggravated contact dermatitis to Kathon CG (methylchloroisothiazolinone/methylisothiazolinone): a novel pattern of involvement in a growing epidemic? | Pirmez, R., et al. 2015. Br J Dermatol. 173: 1343-4. PMID: 26130214
- The fate of two isothiazolinone biocides, 5-chloro-2-methylisothiazol-3(2H)-one (CMI) and 2-methylisothiazol-3(2H)-one (MI), in liquid air fresheners and assessment of inhalation exposure. | Park, SK. and Kwon, JH. 2016. Chemosphere. 144: 2270-6. PMID: 26598996
- Development and validation of an analytical method for determination of bronopol and kathon preservative in milk. | Chilbule, A., et al. 2019. J Food Sci Technol. 56: 3170-3176. PMID: 31205373
- Graphene oxide enhanced ozonation of 5-chloro-2-methyl-4-isothiazolin-3-one: Kinetics, degradation pathway, and toxicity. | Ye, B., et al. 2020. J Hazard Mater. 394: 122563. PMID: 32248031
- Kathon CG: a review. | de Groot, AC. and Weyland, JW. 1988. J Am Acad Dermatol. 18: 350-8. PMID: 3279090
- Occurrence and Transport of Isothiazolinone-Type Biocides from Commercial Products to Aquatic Environment and Environmental Risk Assessment. | Paun, I., et al. 2022. Int J Environ Res Public Health. 19: PMID: 35805435
Inhibitor of:
PDI.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0), 5 g | sc-352647 | 5 g | $94.00 | |||
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0), 25 g | sc-352647A | 25 g | $206.00 | |||
5-Chloro-2-methyl-4-isothiazolin-3-one (CMI/MI > 2.0), 100 g | sc-352647B | 100 g | $501.00 |
