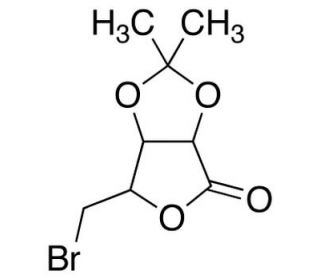
5-Bromo-5-deoxy-2,3-isopropylidene-D-ribonolactone (CAS 94324-23-9)
QUICK LINKS
5-Bromo-5-deoxy-2,3-isopropylidene-D-ribonolactone is a halogenated carbohydrate derivative frequently used in carbohydrate chemistry and glycoscience research. Its bromine substitution at the 5-position and isopropylidene protection of hydroxyl groups make it particularly valuable in synthetic applications, including glycosyl donor and acceptor studies. Researchers employ this compound to probe mechanisms of glycosidic bond formation and stereochemistry. As a glycosyl donor, its 5-bromo group aids in selective glycosylation reactions, allowing scientists to explore the formation of complex oligosaccharides with controlled stereochemistry. Furthermore, its lactone ring and isopropylidene acetal provide stability and a versatile scaffold for further chemical modifications. This compound also serves as a precursor in synthesizing other halogenated carbohydrate derivatives, enabling research into developing inhibitors or probes for studying glycosyltransferase and glycosidase enzymes. By acting as a substrate analog or structural mimic, 5-bromo-5-deoxy-2,3-isopropylidene-D-ribonolactone is crucial in revealing enzyme mechanisms and substrate specificities, particularly in glycosidase activity assays. Additionally, it plays a role in exploring the structural determinants of carbohydrate-binding proteins and enzymes involved in carbohydrate metabolism. Overall, it continues to contribute significantly to synthetic carbohydrate chemistry and enzymology research by facilitating the study of carbohydrate structure, function, and enzyme interactions.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Bromo-5-deoxy-2,3-isopropylidene-D-ribonolactone, 1 g | sc-357847 | 1 g | $268.00 |
