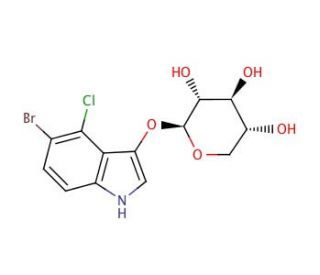
5-Bromo-4-chloro-3-indolyl β-D-xylopyranoside (CAS 207606-55-1)
QUICK LINKS
5-Bromo-4-chloro-3-indolyl β-D-xylopyranoside is a chromogenic substrate used extensively in biochemistry and molecular biology for the detection and analysis of enzyme activity, specifically xylosidases. This compound combines a halogenated indole moiety with a β-D-xylopyranoside group, where the indole ring is modified with bromine and chlorine substituents, enhancing its reactivity and specificity towards enzymatic cleavage. Upon hydrolysis by xylosidases, this substrate releases a halogenated indole, leading to the formation of a colored or fluorescent product that can be easily detected spectroscopically. This characteristic makes it particularly useful for studying the kinetics and mechanism of xylosidases, enzymes that play crucial roles in the degradation of complex carbohydrates and are vital for understanding carbohydrate metabolism in various biological systems. Additionally, it is employed in histochemical applications to localize and quantify xylosidase activity within tissue samples, providing valuable insights into cellular distribution and function of these enzymes. The research use of 5-Bromo-4-chloro-3-indolyl β-D-xylopyranoside is entirely focused on scientific applications, particularly in enhancing our understanding of enzymatic processes within biochemical and cellular contexts.
5-Bromo-4-chloro-3-indolyl β-D-xylopyranoside (CAS 207606-55-1) References
- Heterologous expression and kinetic characterisation of Neurospora crassa β-xylosidase in Pichia pastoris. | Kirikyali, N. and Connerton, IF. 2014. Enzyme Microb Technol. 57: 63-8. PMID: 24629269
- The GH51 α-l-arabinofuranosidase from Paenibacillus sp. THS1 is multifunctional, hydrolyzing main-chain and side-chain glycosidic bonds in heteroxylans. | Bouraoui, H., et al. 2016. Biotechnol Biofuels. 9: 140. PMID: 27398094
- Halotolerant microbial consortia able to degrade highly recalcitrant plant biomass substrate. | Cortes-Tolalpa, L., et al. 2018. Appl Microbiol Biotechnol. 102: 2913-2927. PMID: 29397428
- Screening of multimeric β-xylosidases from the gut microbiome of a higher termite, Globitermes brachycerastes. | Liu, C., et al. 2018. Int J Biol Sci. 14: 608-615. PMID: 29904275
- Modification of DNA regions with metagenomic DNA fragments (MDRMDF): A convenient strategy for efficient protein engineering. | Xu, S., et al. 2021. Biochimie. 187: 75-81. PMID: 34051307
- Immobilization and biochemical properties of a β-xylosidase activated by glucose/xylose from Aspergillus niger USP-67 with transxylosylation activity. | Benassi, Vivian Machado, et al. 2013. Journal of Molecular Catalysis B: Enzymatic. 89: 93-101.
- High salinity tolerant consortia able to growth under a very recalcitrant substrate. | Tolalpa, Larisa Cortés, et al. 2018. Lignocellulose-degrading microbial consortia. 102.6: 95.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Bromo-4-chloro-3-indolyl β-D-xylopyranoside, 25 mg | sc-256898 | 25 mg | $93.00 |
