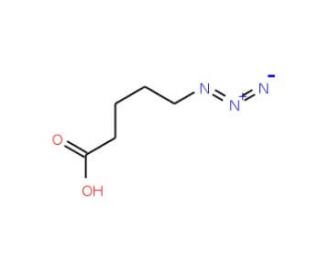
Molecular structure of 5-Azidopentanoic acid, CAS Number: 79583-98-5
5-Azidopentanoic acid (CAS 79583-98-5)
Application:
5-Azidopentanoic acid is a facilitator of the cyclization of peptides.
CAS Number:
79583-98-5
Molecular Weight:
143.14
Molecular Formula:
C5H9N3O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Azidopentanoic acid (5-APA) is a versatile organic acid with extensive applications in scientific research. This compound serves as a fundamental five-carbon precursor in the synthesis of various compounds, including peptides and proteins. Scientific research has harnessed the potential of 5-azidopentanoic acid across a broad spectrum of applications. Its utilization spans the synthesis of peptides and proteins, facilitating investigations into their intricate structure and function. While the precise mechanism of action of 5-azidopentanoic acid remains incompletely understood, current understanding suggests that it binds to specific proteins and enzymes, subsequently activating them.
5-Azidopentanoic acid (CAS 79583-98-5) References
- DOTA derivatives for site-specific biomolecule-modification via click chemistry: synthesis and comparison of reaction characteristics. | Wängler, C., et al. 2011. Bioorg Med Chem. 19: 3864-74. PMID: 21620712
- Coiled coil peptides as universal linkers for the attachment of recombinant proteins to polymer therapeutics. | Pechar, M., et al. 2011. Biomacromolecules. 12: 3645-55. PMID: 21863890
- Cyclopaldic acid, seiridin, and sphaeropsidin A as fungal phytotoxins, and larvicidal and biting deterrents against Aedes aegypti (Diptera: Culicidae): structure-activity relationships. | Cimmino, A., et al. 2013. Chem Biodivers. 10: 1239-51. PMID: 23847068
- Fluorescent probes of the apoptolidins and their utility in cellular localization studies. | DeGuire, SM., et al. 2015. Angew Chem Int Ed Engl. 54: 961-4. PMID: 25430909
- An Integrated Microfluidic Processor for DNA-Encoded Combinatorial Library Functional Screening. | MacConnell, AB., et al. 2017. ACS Comb Sci. 19: 181-192. PMID: 28199790
- Selective Triazenation Reaction (STaR) of Secondary Amines for Tagging Monomethyl Lysine Post-Translational Modifications. | Nwajiobi, O., et al. 2021. Angew Chem Int Ed Engl. 60: 7344-7352. PMID: 33354813
- Transferrin-modified chitosan nanoparticles for targeted nose-to-brain delivery of proteins. | Gabold, B., et al. 2023. Drug Deliv Transl Res. 13: 822-838. PMID: 36207657
- 18F-labeling and initial in vivo evaluation of a Hitomi peptide for imaging tissue transglutaminase 2. | Ackermann, U., et al. 2023. Nucl Med Biol. 116-117: 108308. PMID: 36502585
- Nanoparticles by ROMP in nonaqueous emulsions. | Haschick, R., Klapper, M., Wagener, K. B., & Müllen, K. 2010. Macromolecular Chemistry and Physics. 211(24): 2547-2554.
- Controlling physical properties of iron nanoparticles during assembly by "click chemistry" | Liu, Y., RamaRao, N., Miller, T., Hadjipanayis, G., & Teplyakov, A. V. 2013. The Journal of Physical Chemistry C. 117(39): 19974-19983.
- Click chemistry as a powerful and chemoselective tool for the attachment of targeting ligands to polymer drug carriers | Pola, R., Braunová, A., Laga, R., Pechar, M., & Ulbrich, K. 2014. Polymer Chemistry. 5(4): 1340-1350.
- Cyclo (L-pro-L-tyr), the fungicide isolated from Lysobacter capsici az78: a structure–activity relationship study | Cimmino, A., Puopolo, G., Perazzolli, M., Andolfi, A., Melck, D., Pertot, I., & Evidente, A. 2014. Chemistry of Heterocyclic Compounds. 50: 290-295.
- Template-free assembly of 2D-electrolytes into nanofibres | Marangoni, V. S., Costa, M. C. F., Ng, P. R., Nguyen, H. T. L., Trushin, M., Carvalho, A.,.. & Neto, A. C. 2021. Materials Today Chemistry. 21: 100542.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Azidopentanoic acid, 250 mg | sc-233248 | 250 mg | $220.00 |
