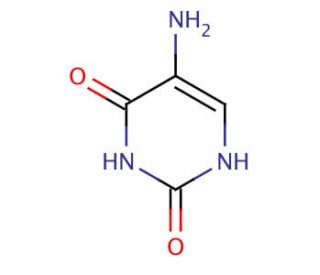
5-Aminouracil (CAS 932-52-5)
QUICK LINKS
5-Aminouracil is a chemical compound that functions as a mutagen in development settings. It exerts its mode of action by incorporating into DNA during replication, leading to base pair mismatches. This results in the induction of mutations, which may be useful for studying DNA replication and repair processes. 5-Aminouracil interferes with the fidelity of DNA synthesis, contributing to the generation of genetic variability in experimental systems. Its ability to induce mutations at specific sites in the genome allows for the investigation of DNA repair mechanisms and the identification of genes involved in maintaining genomic stability. By perturbing the normal DNA replication process, 5-Aminouracil serves as a chemical for understanding the molecular mechanisms underlying genetic diversity and the development of genetics.
5-Aminouracil (CAS 932-52-5) References
- DNA injury induced by 5-aminouracil and caffeine in G2 checkpoints path of higher plant cells. | Del Campo, A., et al. 2005. Biocell. 29: 169-76. PMID: 16187495
- Partial Synchronization of Nuclear Divisions in Root Meristems with 5-Aminouracil. | Smith, HH., et al. 1963. Science. 142: 595-6. PMID: 17738571
- FTIR and raman spectra compared with ab initio calculated frequency modes for 5-aminouracil. | Singh, JS. 2008. J Biol Phys. 34: 569-76. PMID: 19669514
- THE MECHANISM OF 5-AMINOURACIL-INDUCED SYNCHRONY OF CELL DIVISION IN VI CIA FABA ROOT MERISTEMS. | Prensky, W. and Smith, HH. 1965. J Cell Biol. 24: 401-14. PMID: 19866644
- Al3+ selective an efficient colorimetric receptor derived from 5-aminouracil. | Upadhyay, KK. and Kumar, A. 2010. Talanta. 82: 845-9. PMID: 20602979
- Synthesis, reactivity, and biological activity of 5-aminouracil and its derivatives. | Shaker, RM., et al. 2016. Mol Divers. 20: 153-83. PMID: 25926388
- 1-(4-Phenoxybenzyl) 5-Aminouracil Derivatives and Their Analogues - Novel Inhibitors of Human Adenovirus Replication. | Nikitenko, NA., et al. 2018. Acta Naturae. 10: 58-64. PMID: 30116616
- Thermodynamic functions with other properties and vibrational spectra of pyrimidine ring of uracil for RNA and bio-molecule 5-aminouracil. | Singh, JS. 2019. Heliyon. 5: e02955. PMID: 31872124
- 5-Aminouracil and other inhibitors of DNA replication induce biphasic interphase-mitotic cells in apical root meristems of Allium cepa. | Żabka, A., et al. 2020. Plant Cell Rep. 39: 1013-1028. PMID: 32328702
- Structural basis for the interaction modes of dihydroorotase with the anticancer drugs 5-fluorouracil and 5-aminouracil. | Guan, HH., et al. 2021. Biochem Biophys Res Commun. 551: 33-37. PMID: 33714757
- Molecular Insights into How the Dimetal Center in Dihydropyrimidinase Can Bind the Thymine Antagonist 5-Aminouracil: A Different Binding Mode from the Anticancer Drug 5-Fluorouracil. | Lin, ES., et al. 2022. Bioinorg Chem Appl. 2022: 1817745. PMID: 35198016
- 5-Aminouracil treatment. A method for estimating G2. | Socher, SH. and Davidson, D. 1971. J Cell Biol. 48: 248-52. PMID: 5551658
- High mitotic synchronization induced by 5-aminouracil in root cells of Allium cepa L. | Wagenaar, EB. 1966. Exp Cell Res. 43: 184-90. PMID: 5916584
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Aminouracil, 1 g | sc-254769A | 1 g | $13.00 | |||
5-Aminouracil, 5 g | sc-254769 | 5 g | $38.00 | |||
5-Aminouracil, 25 g | sc-254769B | 25 g | $102.00 | |||
5-Aminouracil, 100 g | sc-254769C | 100 g | $326.00 | |||
5-Aminouracil, 500 g | sc-254769D | 500 g | $1311.00 |
