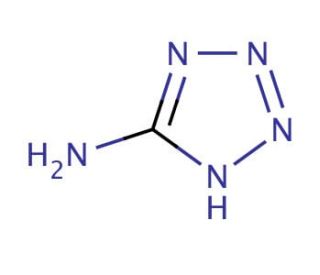
Molecular structure of 5-Aminotetrazole, CAS Number: 4418-61-5
5-Aminotetrazole (CAS 4418-61-5)
CAS Number:
4418-61-5
Purity:
99%
Molecular Weight:
85.07
Molecular Formula:
CH3N5
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Aminotetrazole (5-AT) is a versatile organic compound that belongs to the family of nitrogen-containing heterocyclic compounds. With its white powdery form, 5-Aminotetrazole dissolves readily in water and possesses a melting point of 194-196°C. The applications of 5-Aminotetrazole span various scientific research domains. It finds utility in studying enzyme kinetics and protein folding. Additionally, it plays a significant role in synthesizing diverse organic and inorganic compounds. Although the precise mechanism of action of 5-Aminotetrazole remains not fully elucidated, it is believed to function as a competitive inhibitor of enzymes reliant on a tetrazole moiety for catalysis.
5-Aminotetrazole (CAS 4418-61-5) References
- The mechanism and kinetics of decomposition of 5-aminotetrazole. | Zhang, JG., et al. 2008. J Mol Model. 14: 403-8. PMID: 18330600
- Reaction of 5-aminotetrazole with vinamidinium salts: formation of 2-(N, N-dimethylamino)-5-substituted pyrimidines. | Adnen, HA., et al. 2008. Mol Divers. 12: 61-4. PMID: 18365762
- Unimolecular decomposition of 5-aminotetrazole and its tautomer 5-iminotetrazole: new insight from isopotential searching. | Paul, KW., et al. 2009. J Phys Chem A. 113: 2483-90. PMID: 19236036
- Theoretical study of the 5-aminotetrazole thermal decomposition. | Kiselev, VG. and Gritsan, NP. 2009. J Phys Chem A. 113: 3677-84. PMID: 19323480
- New energetic materials: functionalized 1-ethyl-5-aminotetrazoles and 1-ethyl-5-nitriminotetrazoles. | Stierstorfer, J., et al. 2009. Chemistry. 15: 5775-92. PMID: 19373791
- Phase transition analysis of 5-aminotetrazole from room temperature to the melting point. | Obata, S., et al. 2010. J Phys Chem B. 114: 12572-6. PMID: 20843048
- High-nitrogen-based pyrotechnics: perchlorate-free red- and green-light illuminants based on 5-aminotetrazole. | Sabatini, JJ. and Moretti, JD. 2013. Chemistry. 19: 12839-45. PMID: 23950104
- 5-Aminotetrazole induces spin crossover in iron(III) pentadentate Schiff base complexes: experimental and theoretical investigations. | Herchel, R. and Trávníček, Z. 2013. Dalton Trans. 42: 16279-88. PMID: 24121719
- The reactions of 2-ethoxymethylidene-3-oxo esters and their analogues with 5-aminotetrazole as a way to novel azaheterocycles. | Goryaeva, MV., et al. 2015. Beilstein J Org Chem. 11: 385-91. PMID: 25977712
- In vitro and in vivo effects of 5-aminotetrazole (5-AT), an energetic compound. | Adams, VH., et al. 2020. Regul Toxicol Pharmacol. 111: 104573. PMID: 31884155
- Synthesis of 5-Nitrotetrazolates by the Direct Oxidation of 5-Aminotetrazole in a Single-Pot Synthesis without Isolation of Explosive Intermediates. | Smith, DM., et al. 2020. Chempluschem. 85: 2039-2043. PMID: 32909696
- Mesoionic tetrazolium-5-aminides: Synthesis, molecular and crystal structures, UV-vis spectra, and DFT calculations. | Budevich, VA., et al. 2021. Beilstein J Org Chem. 17: 385-395. PMID: 33633806
- N-Functionalization of 5-Aminotetrazoles: Balancing Energetic Performance and Molecular Stability by Introducing ADNP. | Xiong, J., et al. 2022. Int J Mol Sci. 23: PMID: 36555483
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Aminotetrazole, 100 g | sc-233246 | 100 g | $135.00 |
