5-Amino-1-naphthol (CAS 83-55-6)
QUICK LINKS
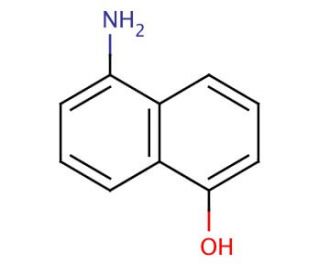
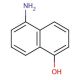
5-Amino-1-naphthol is an organic compound with the chemical formula C10H9NO. It belongs to the class of aromatic compounds known as naphthalenes, which are bicyclic aromatic hydrocarbons. The compound consists of a naphthalene ring system with an amino group (-NH2) attached at the 5-position and a hydroxyl group (-OH) attached at the 1-position.5-Amino-1-naphthol can undergo oxidation reactions, primarily at the hydroxyl group (-OH) attached at the 1-position. Oxidation may involve the transfer of electrons or hydrogen atoms from the compound to an oxidizing agent. This can result in the formation of reactive intermediates or products that can further interact with biological molecules. The amino group (-NH2) attached at the 5-position of 5-Amino-1-naphthol can undergo various chemical reactions. For example, it can participate in nucleophilic substitution reactions, where the amino group acts as a nucleophile and replaces a leaving group in another molecule. This chemical modification can potentially affect the reactivity and interactions of the compound with biological targets. Binding to biological targets: 5-Amino-1-naphthol may interact with various biological targets, such as enzymes, receptors, or DNA. The presence of the amino and hydroxyl groups in the compound can enable hydrogen bonding or other types of interactions with these targets. The binding of the compound to specific biological targets can lead to functional modulation or inhibition of these targets, potentially affecting biological processes.
5-Amino-1-naphthol (CAS 83-55-6) References
- Synthesis and beta 1-, beta 2-adrenergic receptor binding studies of 4-acylamino-substituted phenoxypropanolamine and 5-acylamino-substituted naphthyloxypropanolamine derivatives. | Jindal, DP., et al. 2002. Arzneimittelforschung. 52: 654-63. PMID: 12404879
- Purification and characterization of 1-naphthol-2-hydroxylase from carbaryl-degrading Pseudomonas strain c4. | Swetha, VP., et al. 2007. J Bacteriol. 189: 2660-6. PMID: 17237179
- 5-Amino-1-naphthol: two-dimensional sheets built up from R(4)(4)(18) rings formed by O-H..N, N-H..O and pi-pi interactions. | Rozycka-Sokolowska, E. and Marciniak, B. 2009. Acta Crystallogr C. 65: o565-8. PMID: 19893237
- 5-Amino-1-naphthol, a novel 1,5-naphthalene derivative matrix suitable for matrix-assisted laser desorption/ionization in-source decay of phosphorylated peptides. | Osaka, I., et al. 2013. Rapid Commun Mass Spectrom. 27: 103-8. PMID: 23239322
- Negative Ion In-Source Decay Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry for Sequencing Acidic Peptides. | McMillen, CL., et al. 2016. J Am Soc Mass Spectrom. 27: 847-55. PMID: 26864792
- Photochemical Reactions of Aminonaphthols Caused by Laser Desorption/Ionization. | Nagoshi, K., et al. 2016. Mass Spectrom (Tokyo). 5: A0048. PMID: 27563510
- Characterization of Charge Transfer in Excited States of Extended Clusters of π-Stacked Donor and Acceptor Complexes in Lock-Arm Supramolecular Ordering. | Chen, RX., et al. 2019. J Phys Chem A. 123: 4532-4542. PMID: 31050426
- Nitric oxide permselectivity in electropolymerized films for sensing applications. | Brown, MD. and Schoenfisch, MH. 2016. ACS Sens. 1: 1453-1461. PMID: 31875180
- Influence of Reduced Graphene Oxide on the Electropolymerization of 5-amino-1-naphthol and the Interaction of 1,4-phenylene Diisothiocyanate with the Poly(5-amino-1-naphtol)/Reduced Graphene Oxide Composite. | Baibarac, M., et al. 2020. Polymers (Basel). 12: PMID: 32517061
- Microbial desulfonation of substituted naphthalenesulfonic acids and benzenesulfonic acids. | Zürrer, D., et al. 1987. Appl Environ Microbiol. 53: 1459-63. PMID: 3662502
- Synthesis, characterization, and thermal properties of novel arylene sulfone ether polyimides and polyamides | Barikani, M., & Mehdipour–Ataei, S. 2000. Journal of Polymer Science Part A: Polymer Chemistry. 38(9): 1487-1492.
- Synthesis, characterization, and properties of novel poly (ether urea)s | Mehdipour‐Ataei, S., Sarrafi, Y., & Pirjel, M. A. 2004. Journal of applied polymer science. 93(2): 961-965.
- Preparation of new membranes based on sulfonated aromatic copolyimides | Rabiee, A., Mehdipour‐Ataei, S., Banihashemi, A., & Yeganeh, H. 2008. Polymers for Advanced Technologies. 19(5): 361-370.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Amino-1-naphthol, 25 g | sc-233217 | 25 g | $96.00 | |||
5-Amino-1-naphthol, 100 g | sc-233217A | 100 g | $312.00 |
