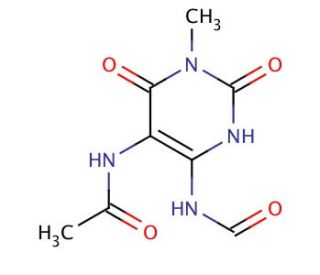
Molecular structure of 5-Acetylamino-6-formylamino-3-methyluracil, CAS Number: 85438-96-6
5-Acetylamino-6-formylamino-3-methyluracil (CAS 85438-96-6)
See product citations (2)
Alternate Names:
AFMU; 5-Acetylamino-6-formylamino-3-methyluracil
Application:
5-Acetylamino-6-formylamino-3-methyluracil is a metabolite of Caffeine
CAS Number:
85438-96-6
Molecular Weight:
226.19
Molecular Formula:
C8H10N4O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-Acetylamino-6-formylamino-3-methyluracil is believed to function by inhibiting the enzyme dihydropyrimidine dehydrogenase (DPD). DPD is responsible for breaking down the nucleoside thymidine. By inhibiting DPD, 5-Acetylamino-6-formylamino-3-methyluracil prevents the breakdown of thymidine, leading to increased levels of thymidine within the cell. This increase in thymidine levels is thought to be the cause of the therapeutic effects of 5-Acetylamino-6-formylamino-3-methyluracil.
5-Acetylamino-6-formylamino-3-methyluracil (CAS 85438-96-6) References
- Validity of an ELISA for N-acetyltransferase-2 (NAT2) phenotyping. | Wong, P., et al. 2001. J Immunol Methods. 251: 1-9. PMID: 11292476
- Stability of 5-acetamido-6-formylamino-3-methyluracil in buffers and urine. | Wong, P., et al. 2002. J Pharm Biomed Anal. 28: 693-700. PMID: 12008149
- Phenotyping of N-acetyltransferase type 2 by caffeine from uncontrolled dietary exposure. | Jetter, A., et al. 2004. Eur J Clin Pharmacol. 60: 17-21. PMID: 14747882
- Re-investigation of the concordance of human NAT2 phenotypes and genotypes. | Bolt, HM., et al. 2005. Arch Toxicol. 79: 196-200. PMID: 15558239
- Measurement of caffeine and five of the major metabolites in urine by high-performance liquid chromatography/tandem mass spectrometry. | Weimann, A., et al. 2005. J Mass Spectrom. 40: 307-16. PMID: 15685651
- In vivo evaluation of CYP1A2, CYP2A6, NAT-2 and xanthine oxidase activities in a Greek population sample by the RP-HPLC monitoring of caffeine metabolic ratios. | Begas, E., et al. 2007. Biomed Chromatogr. 21: 190-200. PMID: 17221922
- Caffeine metabolic ratios for the in vivo evaluation of CYP1A2, N-acetyltransferase 2, xanthine oxidase and CYP2A6 enzymatic activities. | Hakooz, NM. 2009. Curr Drug Metab. 10: 329-38. PMID: 19519341
- Serum Metabolome of Coffee Consumption and its Association With Bone Mineral Density: The Hong Kong Osteoporosis Study. | Chau, YP., et al. 2020. J Clin Endocrinol Metab. 105: PMID: 31750515
- Isolation and identification of 5-acetylamino-6-formylamino-3-methyluracil as a major metabolite of caffeine in man. | Tang, BK., et al. 1983. Drug Metab Dispos. 11: 218-20. PMID: 6135579
- Polymorphic N-acetylation of a caffeine metabolite. | Grant, DM., et al. 1983. Clin Pharmacol Ther. 33: 355-9. PMID: 6825389
- Acetylation phenotypes and biological variation in a French Caucasian population. | Pontes, ZB., et al. 1993. Eur J Clin Chem Clin Biochem. 31: 59-68. PMID: 8467011
- Predominance of slow acetylators of N-acetyltransferase in a Hmong population residing in the United States. | Straka, RJ., et al. 1996. J Clin Pharmacol. 36: 740-7. PMID: 8877679
- Genotype and phenotype of N-acetyltransferase 2 (NAT2) polymorphism in patients with contact allergy. | Schnuch, A., et al. 1998. Contact Dermatitis. 38: 209-11. PMID: 9565293
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-Acetylamino-6-formylamino-3-methyluracil, 5 mg | sc-210263 | 5 mg | $383.00 |
