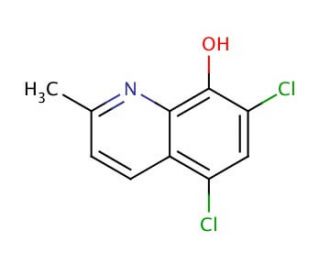
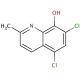
5,7-Dichloro-8-hydroxy-2-methylquinoline (CAS 72-80-0)
QUICK LINKS
5,7-Dichloro-8-hydroxy-2-methylquinoline is a compound that functions as a chelating agent in various experimental applications. Its mechanism of action involves forming stable complexes with metal ions, particularly with transition metals such as iron and copper. This chelation process occurs through the formation of coordinate covalent bonds between the quinoline moiety and the metal ion, leading to the formation of a stable metal complex. 5,7-Dichloro-8-Hydroxy-2-Methylquinoline′s ability to chelate metal ions is utilized in experimental applications to study metal ion homeostasis, metalloprotein function, and metal-catalyzed reactions. 5,7-Dichloro-8-hydroxy-2-methylquinoline is in the development of analytical techniques for the detection and quantification of metal ions in various samples. Its chelating properties make it useful for investigating the role of metal ions in biological systems and for studying metal-mediated processes in chemical reactions.
5,7-Dichloro-8-hydroxy-2-methylquinoline (CAS 72-80-0) References
- 8-Quinolinolato gallium complexes: iso-selective initiators for rac-lactide polymerization. | Bakewell, C., et al. 2013. Inorg Chem. 52: 12561-7. PMID: 24138079
- Structure-activity relationships of anticancer ruthenium(II) complexes with substituted hydroxyquinolines. | Havrylyuk, D., et al. 2018. Eur J Med Chem. 156: 790-799. PMID: 30055464
- Solution Chemistry of Copper(II) Binding to Substituted 8-Hydroxyquinolines. | Summers, KL., et al. 2020. Inorg Chem. 59: 13858-13874. PMID: 32936627
- Copper(II) Binding to PBT2 Differs from That of Other 8-Hydroxyquinoline Chelators: Implications for the Treatment of Neurodegenerative Protein Misfolding Diseases. | Summers, KL., et al. 2020. Inorg Chem. 59: 17519-17534. PMID: 33226796
- A foundational theoreticalAl12E12(E = N, P) adsorption and quinolone docking study: cage-quinolone pairs, optics and possible therapeutic and diagnostic applications. | Ullah, Z., et al. 2023. J Biomol Struct Dyn. 41: 3630-3646. PMID: 35380095
- Controlled Synthesis of Dendrite-like Polyglycerols Using Aluminum Complex for Biomedical Applications. | Perumal, G., et al. 2023. ACS Omega. 8: 2377-2388. PMID: 36687077
- Innovative lanthanide complexes: Shaping the future of cancer/ tumor chemotherapy. | Patyal, M., et al. 2023. J Trace Elem Med Biol. 80: 127277. PMID: 37572546
- Transferrin-inspired iron delivery across the cell membrane using [(L2Fe)2(μ-O)] (L = chlorquinaldol) to harness anticancer activity of ferroptosis. | Abeydeera, N., et al. 2024. Dalton Trans. 53: 3206-3214. PMID: 38247554
- Polymorphism and color dimorphism of chlorquinaldol (5,7-dichloro-8-hydroxy-2-methylquinoline). | Pavlova, AV., et al. 1985. Pharmazie. 40: 730. PMID: 4080791
- Synthesis, Structure, and Antiproliferative Activities of Complexes of Some Transition Metals with 5, 7-Dichloro-8-Hydroxyquinoline-2-Carboxaldehyde-4-Phenyl-3-Thiosemicarbazone | Linh, P. T. H., Chi, M. P., Thao, L. P., Giang, N. T. M., & Hai, L. T. H. 2024. Pharmaceutical Chemistry Journal. 57: 1593–1598.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5,7-Dichloro-8-hydroxy-2-methylquinoline, 10 g | sc-239082 | 10 g | $61.00 |
