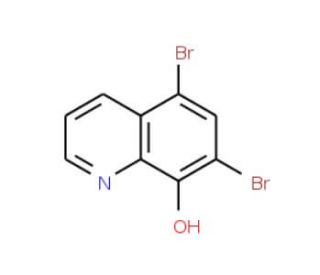
Molecular structure of 5,7-Dibromo-8-hydroxyquinoline, CAS Number: 521-74-4
5,7-Dibromo-8-hydroxyquinoline (CAS 521-74-4)
Alternate Names:
5,7-Dibromo-8-quinolinol; Broxyquinoline; 5,7-Dibromooxine; Bromoxine
CAS Number:
521-74-4
Purity:
≥96%
Molecular Weight:
302.95
Molecular Formula:
C9H5Br2NO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5,7-Dibromo-8-hydroxyquinoline, an organic compound, holds significant utility in the scientific domain, finding multiple applications as a chelating agent, corrosion inhibitor, and a ligand for metal ions. Its versatility extends to a diverse array of scientific research applications. Moreover, it is deemed to exhibit antioxidant properties, shielding cells from oxidative harm. Additionally, this compound is thought to function as an enzyme inhibitor, effectively hindering specific catalytic reactions.
5,7-Dibromo-8-hydroxyquinoline (CAS 521-74-4) References
- A rapid spectrophotometric method for the determination of molybdenum in industrial, environmental, biological and soil samples using 5,7-dibromo-8-hydroxyquinoline. | Ahmed, MJ. and Haque, ME. 2002. Anal Sci. 18: 433-9. PMID: 11999518
- A simple spectrophotometric method for the determination of cadmium in industrial, environmental, biological and soil samples using 5,7-dibromo-8-hydroxyquinoline. | Ahmed, MJ. and Chowdhury, MT. 2004. Anal Sci. 20: 987-90. PMID: 15228125
- The evaluation of liposome-water partitioning of 8-hydroxyquinolines and their copper complexes. | Kaiser, SM. and Escher, BI. 2006. Environ Sci Technol. 40: 1784-91. PMID: 16570598
- Extraction of tungsten with 8-hydroxyquinoline and some of its derivatives. | Awad, K., et al. 1971. Talanta. 18: 279-85. PMID: 18960884
- Synthesis and characterization of 8-hydroxyquinoline complexes of tin(IV) and their application in organic light emitting diode. | Fazaeli, Y., et al. 2012. J Fluoresc. 22: 1263-70. PMID: 22695927
- New record of Scedosporium dehoogii from Chile: Phylogeny and susceptibility profiles to classic and novel putative antifungal agents. | Alvarez, E. and Sanhueza, C. 2016. Rev Iberoam Micol. 33: 224-229. PMID: 27461998
- Uptake of 63Ni2+ in the central and peripheral nervous system of mice after oral administration: effects of treatments with halogenated 8-hydroxyquinolines. | Borg, K. and Tjälve, H. 1989. Toxicology. 54: 59-68. PMID: 2916242
- Decision making for promising quinoline-based anticancer agents through combined methodology. | Özcan, E., et al. 2020. J Biochem Mol Toxicol. e22522. PMID: 32407595
- Quinoline-based promising anticancer and antibacterial agents, and some metabolic enzyme inhibitors. | Ökten, S., et al. 2020. Arch Pharm (Weinheim). 353: e2000086. PMID: 32537757
- PBT2 acts through a different mechanism of action than other 8-hydroxyquinolines: an X-ray fluorescence imaging study. | Summers, KL., et al. 2020. Metallomics. 12: 1979-1994. PMID: 33169753
- Alzheimer's Drug PBT2 Interacts with the Amyloid β 1-42 Peptide Differently than Other 8-Hydroxyquinoline Chelating Drugs. | Summers, KL., et al. 2022. Inorg Chem. 61: 14626-14640. PMID: 36073854
- A Novel Family of Cage-like (CuLi, CuNa, CuK)-phenylsilsesquioxane Complexes with 8-Hydroxyquinoline Ligands: Synthesis, Structure, and Catalytic Activity. | Bilyachenko, AN., et al. 2022. Molecules. 27: PMID: 36234735
- Non-extractive spectrophotometric determination of vanadium(v) in alloys and environmental, biological and soil samples using 5,7-dibromo-8-hydroxyquinoline. | Ahmed, MJ. and Banerjee, AK. 1995. Analyst. 120: 2019-23. PMID: 7661342
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5,7-Dibromo-8-hydroxyquinoline, 50 g | sc-352899 | 50 g | $60.00 | |||
5,7-Dibromo-8-hydroxyquinoline, 250 g | sc-352899A | 250 g | $246.00 |
