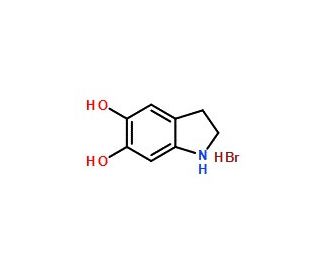
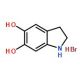
5,6-Dihydroxyindoline hydrobromide (CAS 29539-03-5)
QUICK LINKS
5,6-Dihydroxyindoline hydrobromide, also known as IDH or indoline-5,6-diol hydrobromide, is an extensively utilized organic compound in the domains of organic synthesis, pharmaceuticals, and biochemistry. Serving as a foundational component for synthesizing diverse compounds, 5,6-Dihydroxyindoline hydrobromide finds widespread applications across multiple fields. It manifests as a colorless, crystalline solid with hygroscopic properties and demonstrates solubility in water and ethanol. Alternate names for 5,6-Dihydroxyindoline hydrobromide include 5-bromo-4-indoline-2-diol or 5-bromo-4-hydroxy-2-indoline. Researchers propose that 5,6-Dihydroxyindoline hydrobromide operates as an inhibitor of enzymes, effectively impeding their activity. Moreover, 5,6-Dihydroxyindoline hydrobromide exhibits antioxidant qualities by effectively scavenging free radicals and providing protection against oxidative damage.
5,6-Dihydroxyindoline hydrobromide (CAS 29539-03-5) References
- Bienzymatic electrode for the determination of aspartame in dietary products. | Fatibello-Filho, O., et al. 1988. Anal Chem. 60: 2397-9. PMID: 3239806
- Studies related to the chemistry of melanins. V. Investigations on the specific deuteriation of 5,6-dihydroxyindoline and 5,6-dihydroxy-indole. | Mishra, SN. and Swan, GA. 1967. J Chem Soc Perkin 1. 15: 1431-5. PMID: 6068311
- Generation of the neurotoxin 6-hydroxydopamine by peroxidase/H2O2 oxidation of dopamine. | Napolitano, A., et al. 1995. J Med Chem. 38: 917-22. PMID: 7699708
- Iron-mediated generation of the neurotoxin 6-hydroxydopamine quinone by reaction of fatty acid hydroperoxides with dopamine: a possible contributory mechanism for neuronal degeneration in Parkinson's disease. | Pezzella, A., et al. 1997. J Med Chem. 40: 2211-6. PMID: 9216840
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5,6-Dihydroxyindoline hydrobromide, 250 mg | sc-482222 | 250 mg | $316.00 | |||
5,6-Dihydroxyindoline hydrobromide, 1 g | sc-482222A | 1 g | $618.00 |
