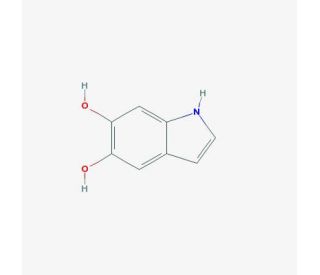
Molecular structure of 5,6-Dihydroxyindole, CAS Number: 3131-52-0
5,6-Dihydroxyindole (CAS 3131-52-0)
See product citations (1)
Alternate Names:
1H-Indole-5,6-diol; Dopamine lutine; Indole-5,6-diol
Application:
5,6-Dihydroxyindole is a melanin precursor
CAS Number:
3131-52-0
Purity:
≥97%
Molecular Weight:
149.15
Molecular Formula:
C8H7NO2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5,6-Dihydroxyindole is a occurring compound found in various biological systems. 5,6-Dihydroxyindole is a key intermediate in the melanin synthesis pathway, where it undergoes further chemical reactions to form the final pigment. It plays a role in determining the coloration of tissues and provides protection against the harmful effects of ultraviolet radiation. 5,6-Dihydroxyindole has been studied for its potential antioxidant properties, which may have implications for cellular protection and defense against oxidative stress.
5,6-Dihydroxyindole (CAS 3131-52-0) References
- Fe(III)-coordination properties of neuromelanin components: 5,6-dihydroxyindole and 5,6-dihydroxyindole-2-carboxylic acid. | Charkoudian, LK. and Franz, KJ. 2006. Inorg Chem. 45: 3657-64. PMID: 16634598
- Short-lived quinonoid species from 5,6-dihydroxyindole dimers en route to eumelanin polymers: integrated chemical, pulse radiolytic, and quantum mechanical investigation. | Pezzella, A., et al. 2006. J Am Chem Soc. 128: 15490-8. PMID: 17132016
- The first 5,6-dihydroxyindole tetramer by oxidation of 5,5',6,6'-tetrahydroxy- 2,4'-biindolyl and an unexpected issue of positional reactivity en route to eumelanin-related polymers. | Panzella, L., et al. 2007. Org Lett. 9: 1411-4. PMID: 17346057
- 5,6-dihydroxyindole tetramers with 'anomalous' interunit bonding patterns by oxidative coupling of 5,5',6,6'-tetrahydroxy-2,7'-biindolyl: emerging complexities on the way toward an improved model of eumelanin buildup. | Pezzella, A., et al. 2007. J Org Chem. 72: 9225-30. PMID: 17975929
- π-Electron manipulation of the 5,6-dihydroxyindole/quinone system by 3-alkynylation: mild acid-mediated entry to (cross)-conjugated scaffolds and paradigms for medium-tunable chromophores. | Capelli, L., et al. 2011. J Org Chem. 76: 4457-66. PMID: 21539372
- UV-absorption spectra of melanosomes containing varying 5,6-dihydroxyindole and 5,6-dihydroxyindole-2-carboxylic acid content. | Peles, DN. and Simon, JD. 2011. J Phys Chem B. 115: 12624-31. PMID: 21923179
- Towards eumelanin@zeolite hybrids: pore-size-controlled 5,6-dihydroxyindole polymerization. | Prasetyanto, EA., et al. 2014. Chemistry. 20: 1597-601. PMID: 24403183
- Degree of polymerization of 5,6-dihydroxyindole-derived eumelanin from chemical degradation study. | Okuda, H., et al. 2014. Pigment Cell Melanoma Res. 27: 664-7. PMID: 24750564
- Direct Evidence for the Critical Role of 5,6-Dihydroxyindole in Polydopamine Deposition and Aggregation. | Lyu, Q., et al. 2019. Langmuir. 35: 5191-5201. PMID: 30916980
- Dynamics of electronically excited states in the eumelanin building block 5,6-dihydroxyindole. | Crane, SW., et al. 2019. Phys Chem Chem Phys. 21: 8152-8160. PMID: 30933211
- Nonenzymatic Spontaneous Oxidative Transformation of 5,6-Dihydroxyindole. | Sugumaran, M., et al. 2020. Int J Mol Sci. 21: PMID: 33023030
- Preparation of eumelanin-related metabolites 5,6-dihydroxyindole, 5,6-dihydroxyindole-2-carboxylic acid, and their O-methyl derivatives. | Wakamatsu, K. and Ito, S. 1988. Anal Biochem. 170: 335-40. PMID: 3394933
- Analogs of the Dopamine Metabolite 5,6-Dihydroxyindole Bind Directly to and Activate the Nuclear Receptor Nurr1. | Kholodar, SA., et al. 2021. ACS Chem Biol. 16: 1159-1163. PMID: 34165961
- 5,6-Dihydroxyindole eumelanin content in human skin with varying degrees of constitutive pigmentation. | Del Bino, S., et al. 2022. Pigment Cell Melanoma Res. 35: 622-626. PMID: 35933709
- Hydrogen peroxide generation associated with the oxidations of the eumelanin precursors 5,6-dihydroxyindole and 5,6-dihydroxyindole-2-carboxylic acid. | Nappi, AJ. and Vass, E. 1996. Melanoma Res. 6: 341-9. PMID: 8908594
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5,6-Dihydroxyindole, 250 mg | sc-280510 | 250 mg | $235.00 | |||
5,6-Dihydroxyindole, 1 g | sc-280510A | 1 g | $352.00 |
