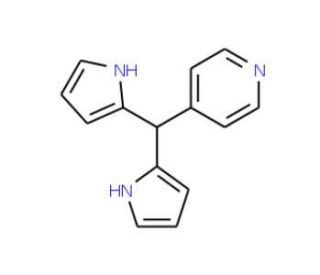
Molecular structure of 5-(4-Pyridyl)dipyrromethane, CAS Number: 52073-75-3
5-(4-Pyridyl)dipyrromethane (CAS 52073-75-3)
Alternate Names:
4-(DI-PYRROL-2-YL-METHYL)PYRIDINE
Application:
5-(4-Pyridyl)dipyrromethane is a pyrrole building block for porphyrin synthesis
CAS Number:
52073-75-3
Molecular Weight:
223.27
Molecular Formula:
C14H13N3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
5-(4-Pyridyl)dipyrromethane is a highly versatile compound with numerous applications in scientific research. 5-(4-Pyridyl)dipyrromethane is a pyrrole building block for porphyrin synthesis. Its unique properties have led to investigations into its potential as an antioxidant, making it valuable for studying oxidative stress and related processes. Additionally, this compound shows promise as a photosensitizer in photodynamic therapy, where it can help facilitate the targeted destruction of cancer cells using light. Furthermore, its ability to efficiently harvest and transfer energy makes it a promising candidate for use in organic solar cells, contributing to advancements in renewable energy technologies.
5-(4-Pyridyl)dipyrromethane (CAS 52073-75-3) References
- Rational synthesis of meso-substituted porphyrins bearing one nitrogen heterocyclic group. | Gryko, D. and Lindsey, JS. 2000. J Org Chem. 65: 2249-52. PMID: 10774058
- Heteroleptic copper dipyrromethene complexes: synthesis, structure, and coordination polymers. | Halper, SR., et al. 2004. Inorg Chem. 43: 1242-9. PMID: 14966958
- A survey of acid catalysis and oxidation conditions in the two-step, one-flask synthesis of meso-substituted corroles via dipyrromethanedicarbinols and pyrrole. | Geier, GR., et al. 2004. J Org Chem. 69: 4159-69. PMID: 15176843
- Investigation of stepwise covalent synthesis on a surface yielding porphyrin-based multicomponent architectures. | Schmidt, I., et al. 2006. J Org Chem. 71: 3033-50. PMID: 16599598
- Topological control in heterometallic metal-organic frameworks by anion templating and metalloligand design. | Halper, SR., et al. 2006. J Am Chem Soc. 128: 15255-68. PMID: 17117878
- Rare examples of transition-metal-main-group metal heterometallic metal-organic frameworks from gallium and indium dipyrrinato complexes and silver salts: synthesis and framework variability. | Stork, JR., et al. 2007. Inorg Chem. 46: 11213-23. PMID: 18047326
- A pared-down version of 5,10,15,20-tetra(N-methylpyridinium-4-yl)porphyrin intercalates into B-form DNA regardless of base composition: binding studies of tri(N-methylpyridinium-4-yl)porphyrins. | Andrews, K. and McMillin, DR. 2008. Biochemistry. 47: 1117-25. PMID: 18171084
- Pd(II)-mediated assembly of porphyrin channels in bilayer membranes. | Devi, U., et al. 2011. Langmuir. 27: 1448-56. PMID: 21174428
- Fine tuning the reactivity of corrole-based catalytic antioxidants. | Okun, Z. and Gross, Z. 2012. Inorg Chem. 51: 8083-90. PMID: 22808919
- Rational design of fluorophores for in vivo applications. | Ptaszek, M. 2013. Prog Mol Biol Transl Sci. 113: 59-108. PMID: 23244789
- Photoinduced antibacterial activity of two dicationic 5,15-diarylporphyrins. | Orlandi, VT., et al. 2013. J Photochem Photobiol B. 127: 123-32. PMID: 24041850
- The effect of pyridyl substituents on the thermodynamics of porphyrin binding to G-quadruplex DNA. | Rowland, GB., et al. 2013. Bioorg Med Chem. 21: 7515-22. PMID: 24148836
- Imprinting Pentaphyrin on Conductive Electropolymerized Dipyrromethane Films: A New Strategy towards the Synthesis of Photokilling Materials. | Comuzzi, C., et al. 2020. Chempluschem. 85: 776-782. PMID: 32202701
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
5-(4-Pyridyl)dipyrromethane, 250 mg | sc-262302 | 250 mg | $428.00 | |||
5-(4-Pyridyl)dipyrromethane, 1 g | sc-262302A | 1 g | $434.00 |
